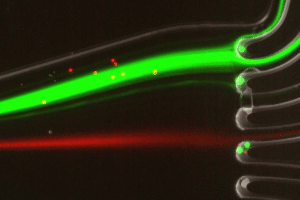
Contamination-free, sterile
Organ on Chip Perfusion Pack
The perfect set for organ on chip perfusion.
The perfect setup for liquid handling in Organ on Chip experiments.
Fluigent has joined with Beonchip to offer complete solutions for organ-on-chip applications combining innovative, robust, and easy-to-use chips with high-performance instruments.
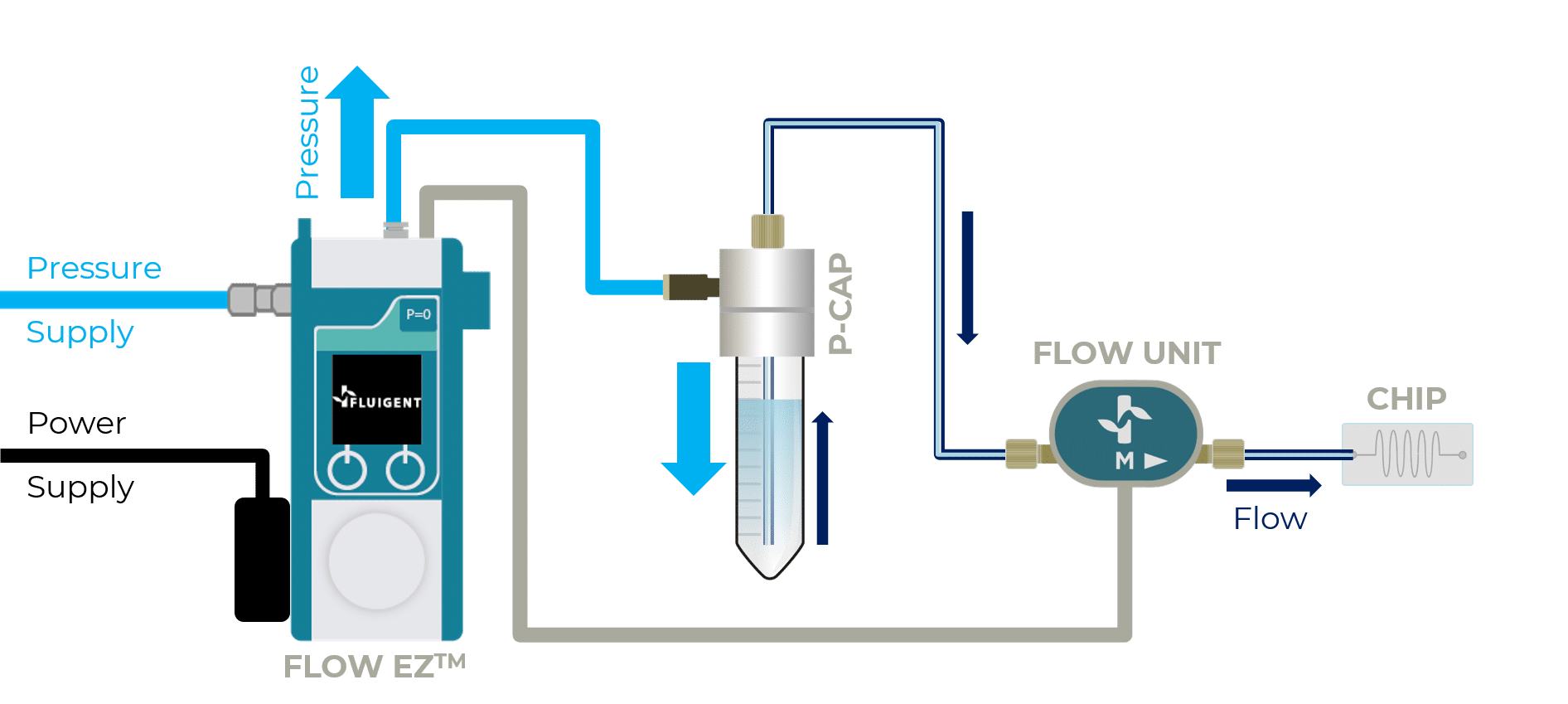
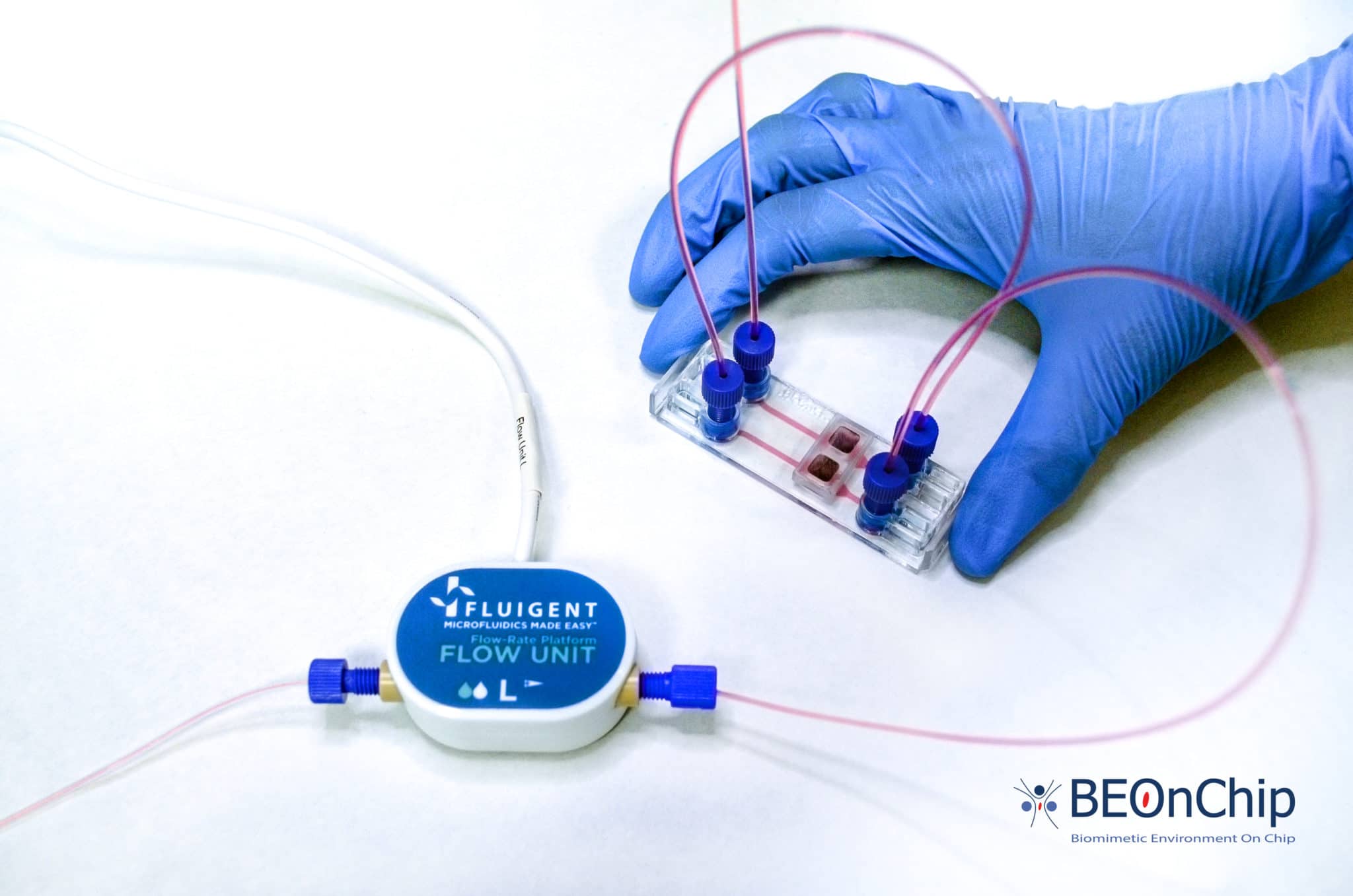
This Organ on Chip Perfusion Pack allows users to obtain stable medium perfusion through the controlled and stable flow of multiple solutions into the microfluidic chip, combining a bidirectional microfluidic flow sensor and a microfluidic pressure-based flow controller. Users can perform and monitor their organ-on-a-chip experiment using our OxyGEN software interface. The software automates the experiment and fine-tunes the flow parameters.

- Designed for cell biology
- Easy to use
Simple connections
Features of the OOC Perfusion Pack
Optimized seeding
Cell seeding in 3D hydrogel presents challenges.. Hydrogels are fragile and can easily fracture or detach from the chip if the flow is unstable.
Manual cell seeding is not recommended as the discontinuous flow results in heterogeneous cell seeding and can crack the hydrogel.
The manual mode of the Flow EZ included in the Organ on Chip Perfusion Pack is perfect to circumvent these limitations as it allows the implementation of small flow rate increments while keeping an eye on the microscope.
Generate complex flow patterns
Control flow rate or pressure for applications with constant shear stress such as vascular perfusion.
Reproduce complex flow patterns such as aortic pressure variations while maintaining a unidirectional flow in the chip.
Protocol automation
Once the parameters are set and optimized, protocol automation saves time, limits contamination, and decreases variability.
Valves, pressure, and flow controllers from Fluigent can be assembled to automate any protocol using user-friendly software (OxyGEN).
Customization possible
Fluigent’s microfluidic perfusion pack is customizable to fit the needs of specific flow rates and pressures.
Main products of the Organ on Chip Pack

Microfluidic flow controller
Flow EZ™
Read more
Bidirectional Microfluidic Flow Sensor
FLOW UNIT | FLOW UNIT +
Read more
Microfluidic cell culture chip
Be-Flow perfused cell culture chip
Read more
Double channel Microfluidic chip for hypoxic cell culture
BE-DoubleFlow
Read more
Air Liquid Interface Cell culture versatile chip
Be-Transflow
Read more
Airtight metal tube caps for microfluidics
P-CAP series
Read more
Why use this microfluidic perfusion pack?
Organ-on-chip studies are gauging interest from the scientific community due to their ability to mimic an in-vivo environment in a precise and controlled manner. In particular, introducing shear stress through flow control and reproducing biochemical environments in cell cultures was made possible due to advances in the organ on chip technologies.
The Organ on Chip Perfusion Pack will allow users to control both pressure and flow rate, switch between media and drugs, obtain a stable and pulseless flow rate, have a large range of flow rates, and customize and create complex patterns.
By combining microfluidic technology, micromachining, and cell biology, environmental parameters can be precisely controlled on organs-on-chips. This may generate fluid shear stress, mechanical stress, biochemical concentration gradients and other physical and chemical stimuli. The cells on the chip can respond to these stimuli, develop self-organization, and express more realistic physiological functions like exhibiting special advantages in the establishment of an in vitro physiological model.
Cell culture under flow
Cultivating cells under flow perfusion has two main advantages: constant medium renewal and reproduction of mechanical strain in vitro. To grow cells under perfusion using a cell perfusion pack, we recommend using the BE-Flow chip.

Chemotaxis
Cells are constantly exposed to biochemical stimulation from the early embryonic stage to adult life. The spatiotemporal regulation of these signals is essential as it determines cell fate, phenotype, metabolic activity as well as pathological behaviors. Biochemical stimulation is also central in oncology as it drives the migration and expansion of malign tumors.
To study chemotaxis in 3D hydrogel we recommend using BE-GRADIENT with the Organ on Chip Perfusion Pack.
Co-culture under perfusion
Perform co-culture under perfusion with our Organ on Chip Pack, which is essential for various applications for the following reasons:
- To fully differentiate given cell types, coculture with another cell type is required
- In cosmetics or drug testing to reproduce interfaces link skin, lung or gut and monitor the uptake of active molecules
- To reproduce liquid-liquid or air-liquid interfaces.
The Be-Doubleflow chip best fit applications in organ-on-chip cell perfusion as a porous membrane separates the central chamber into two channels.

Transwell under flow
Transwells are classic devices widely used in standard cell culture to co-culture cell types, reproduce 2D-2D interfaces, 3D-2D interfaces, study cell invasion, assess monolayer permeability or active molecule uptake.
However, Transwell devices use large volumes of liquid, and communication is therefore slow and low concentration, signalling factors are diluted, which altogether hampers studying cellular communication. Furthermore, the culture is entirely static, which precludes emulation of dynamic processes and the application of controlled cell biochemical and/or physical stimuli.
Using a microfluidic format can solve some of these issues by offering sub-milliliter volumes, dynamic culture, and exquisite spatiotemporal control over physical and chemical parameters in the cell/tissue vicinity.
For this application, we recommend the Be-Transflow included in the Organ on Chip Perfusion Pack.
Cell culture with regulated Shear Stress
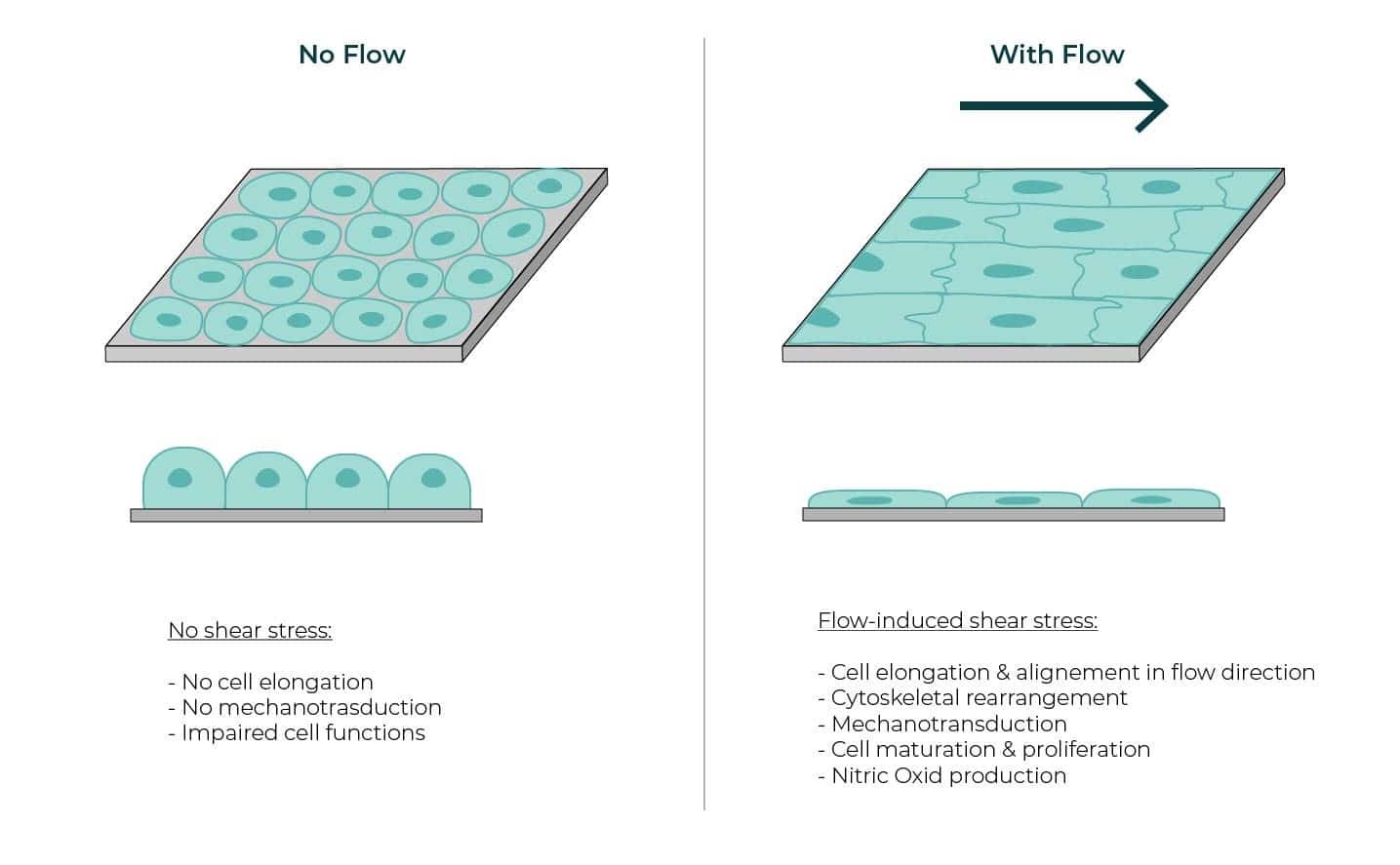
Mechanical forces are potent regulators of cellular structures and functions in both health and disease. As a result of their unique location, endothelial cells experience several mechanical forces.
Of these forces, shear stress is particularly important as it stimulates the release
of vasoactive substances and changes gene expression, cell metabolism, and cell morphology.
To grow cells under regulated shear stress, we recommend to use the OOC perfusion pack with the Be-FLOW
| No shear stress | Flow-induced shear stress |
| – No cell elongation – No mechanotransduction – Impaired cell functions | – Cell elongation & alignement in flow direction -Cytoskeletal rearrangement – Mechanotransduction – Cell maturation & proliferation – Nitric Oxid production |
Specifications
| LineUp Flow EZ pressure controller |
| LineUp LINK Module (software control) |
| Beonchip microfluidic chip |
| Fluigent connectors and tubing kit |
OxyGEN
| Control in real-time, protocol automation, data record and export |
| ver. 2.2.0.0 or more recent |
Software Development Kit
| Custom software application |
| ver. 22.2.0.0 or more recent |
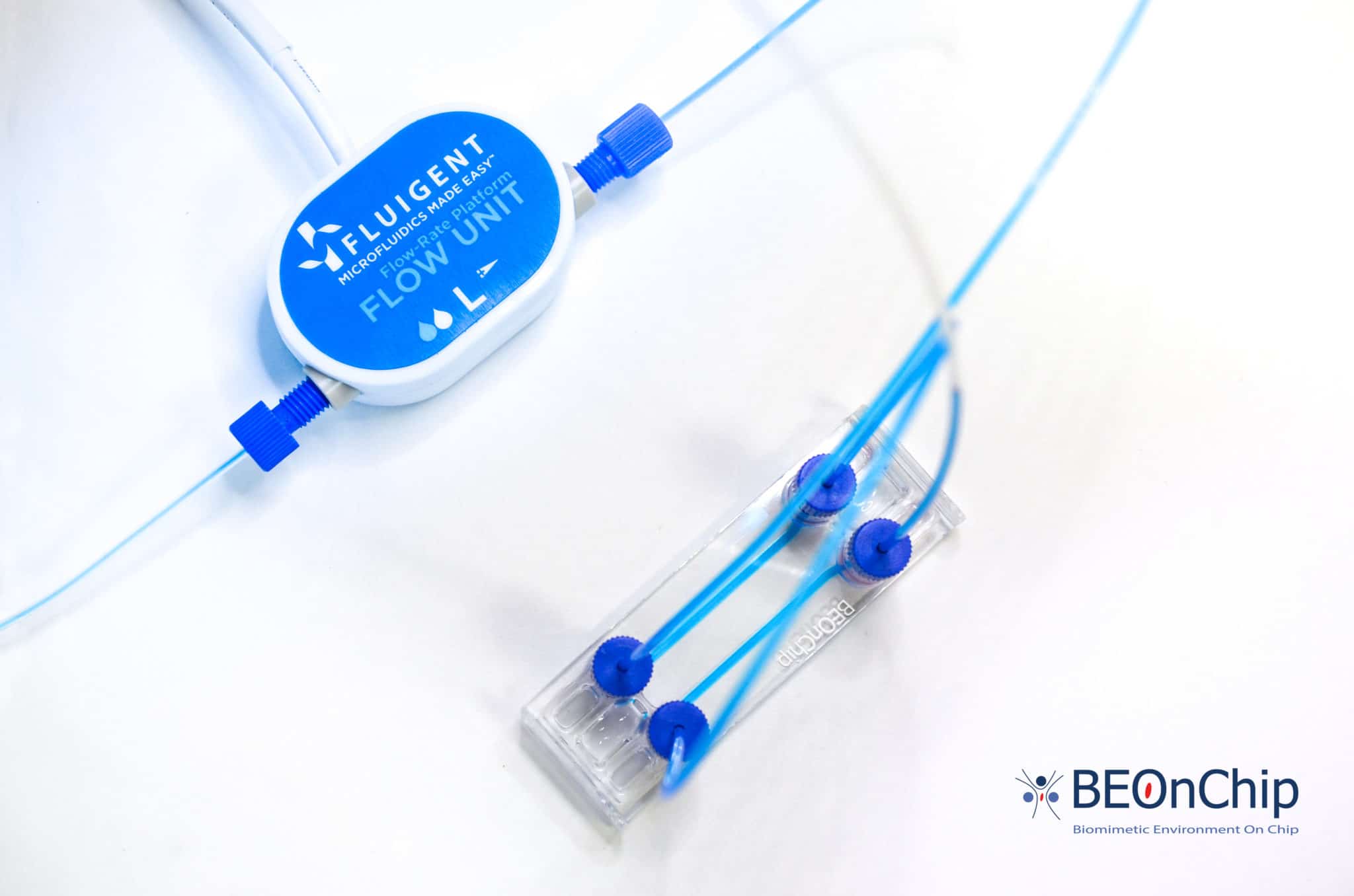
Connect the chip to the microfluidic system
Switching from static culture to fluid perfusion
Expertise & resources
-
Expert Reviews: Basics of Microfluidics Why Control Shear Stress in Cell Biology? Read more
-
Microfluidic Application Notes Development of a human gut-on-chip to assess the effect of shear stress on intestinal functions Read more
-
Microfluidic Application Notes Peristaltic Pump vs Pressure-Based Microfluidic Flow Control for Organ on Chip applications Read more
-
Fluigent Products Datasheets BE-Gradient datasheet Download
-
Fluigent Products Datasheets BE-Transflow datasheet Download
-
Fluigent Products Datasheets BE-flow datasheet Download
-
Fluigent Products Datasheets BE-DoubleFlow Standard datasheet Download
-
Expert Reviews: Basics of Microfluidics Passive and active mechanical stimulation in microfluidic systems Read more
-
Fluigent Products Datasheets FLOW UNIT Datasheet Download
-
Expert Reviews: Basics of Microfluidics Mimicking in-vivo environments: biochemical and biomechanical stimulation Read more
-
Fluigent Products Datasheets Flow EZ™ Datasheet Download