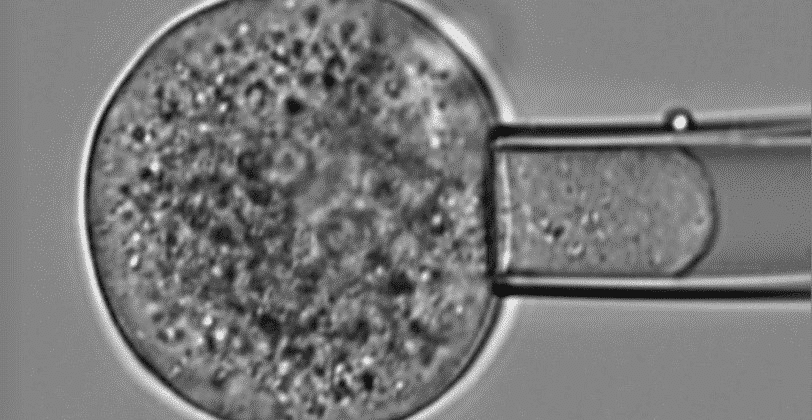
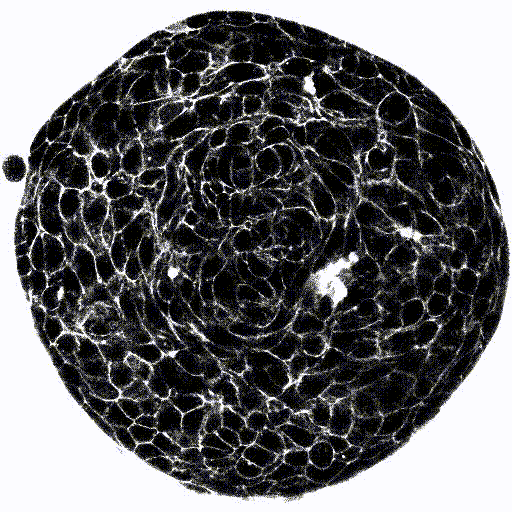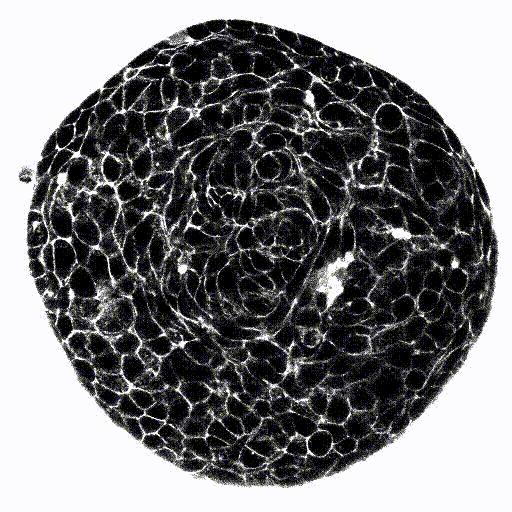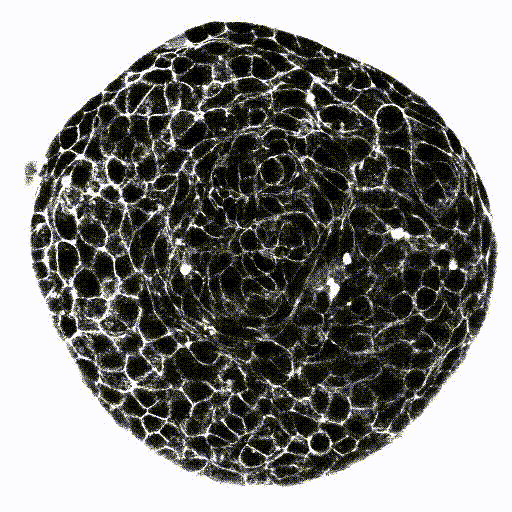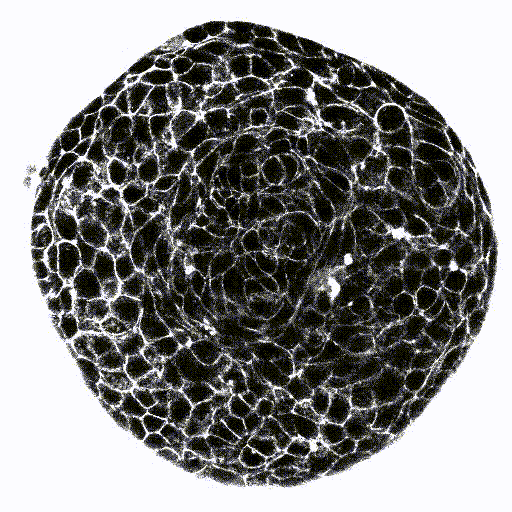
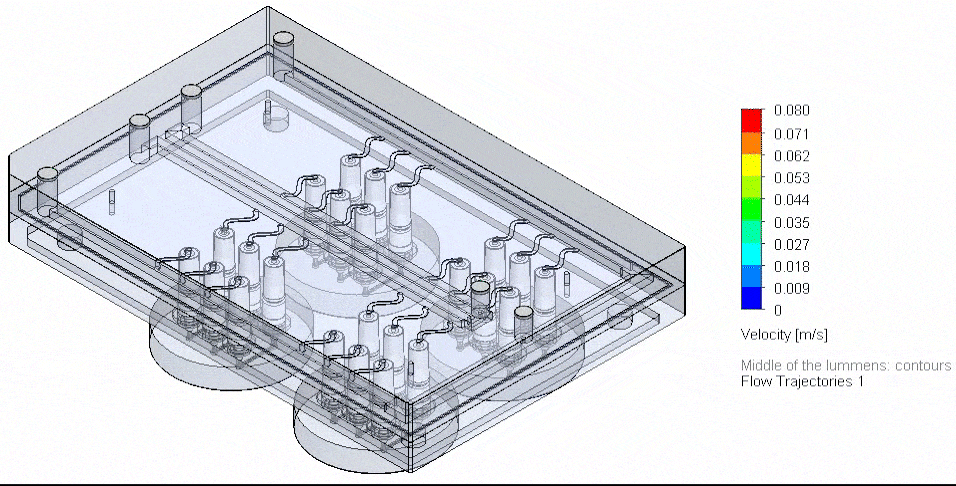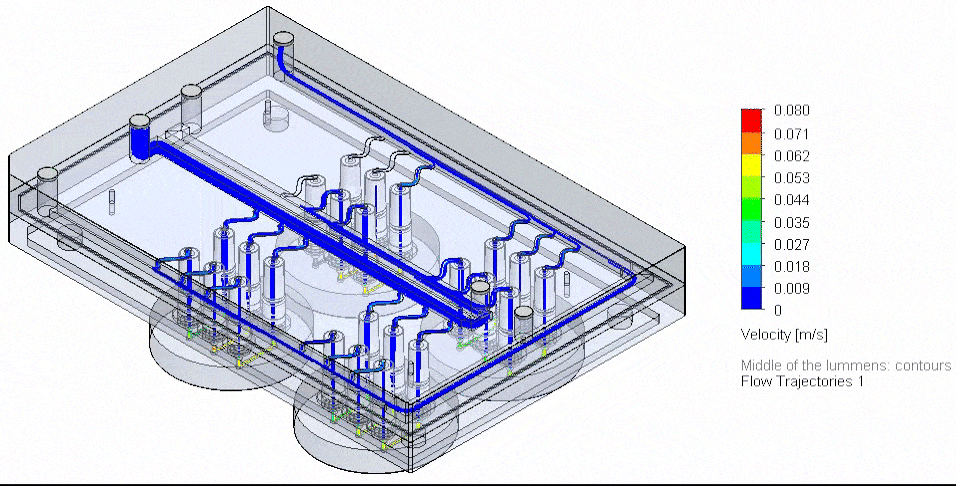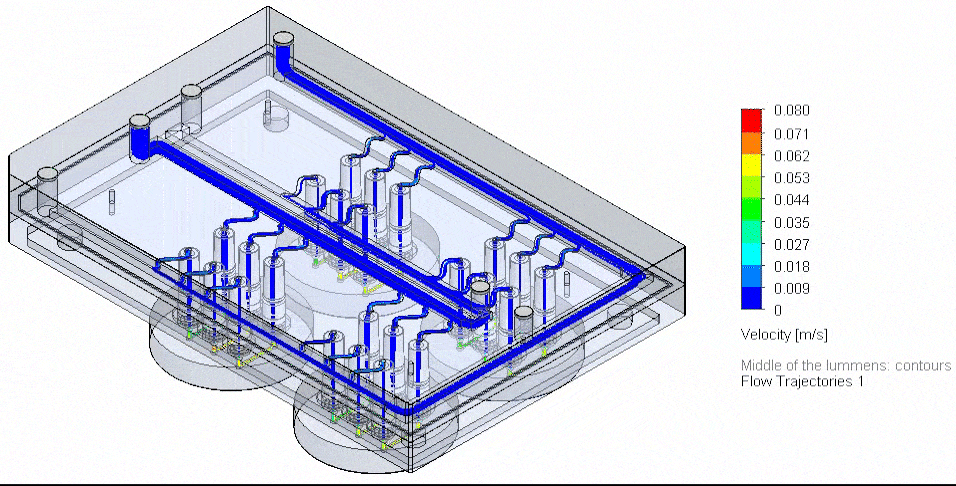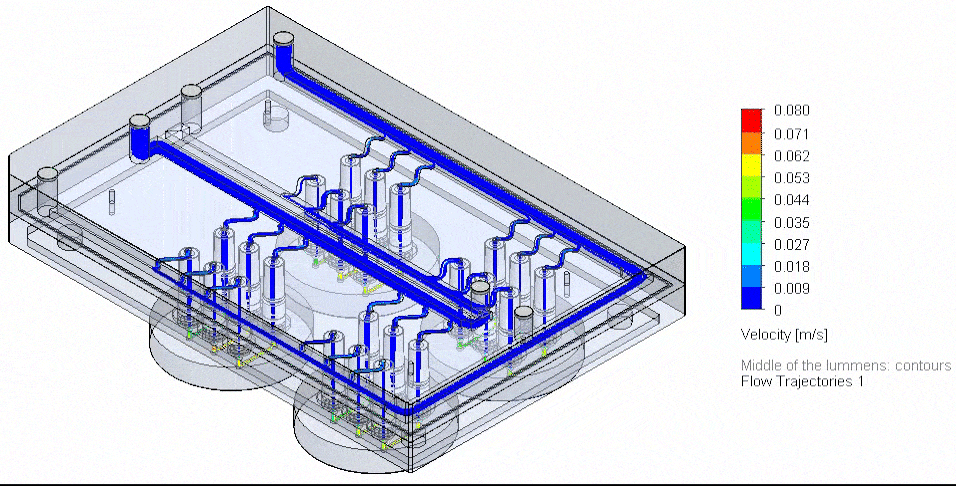
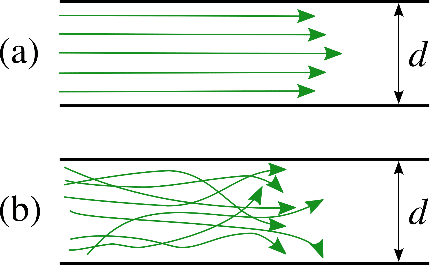
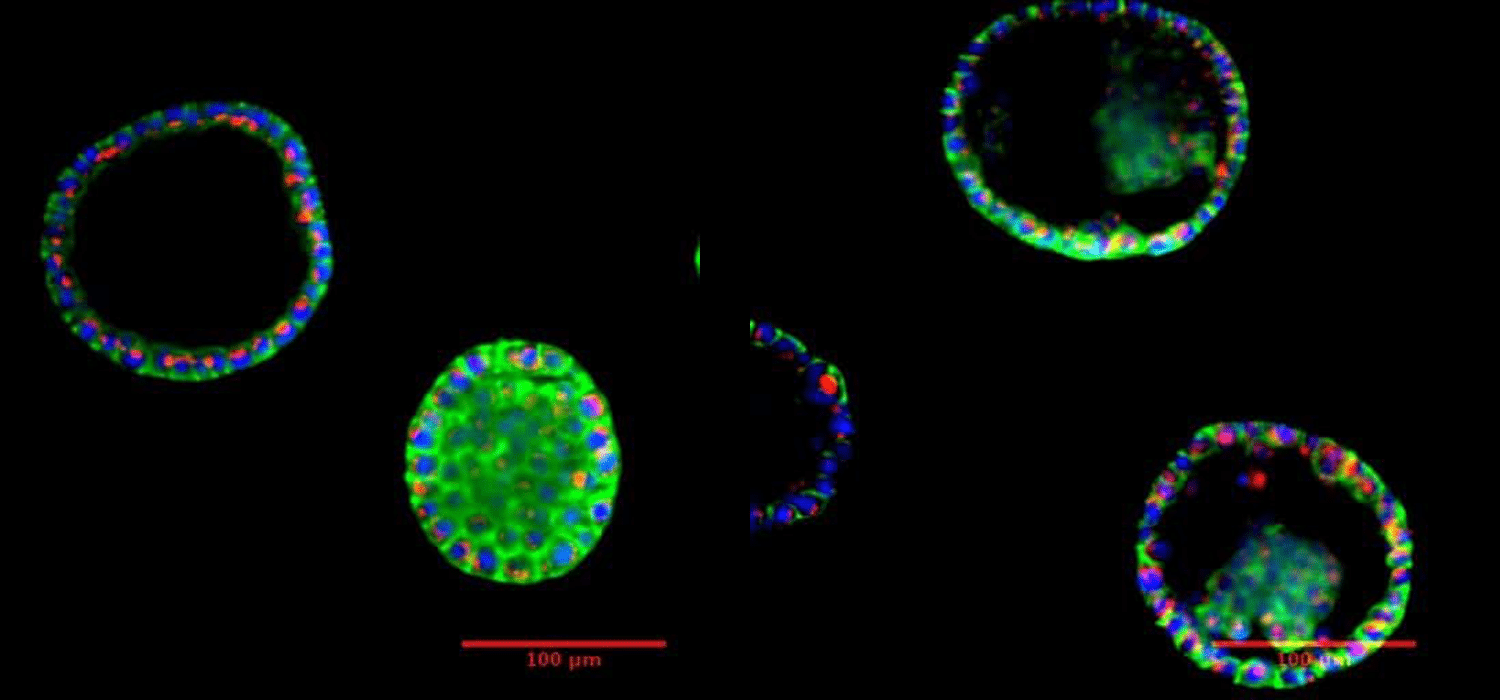
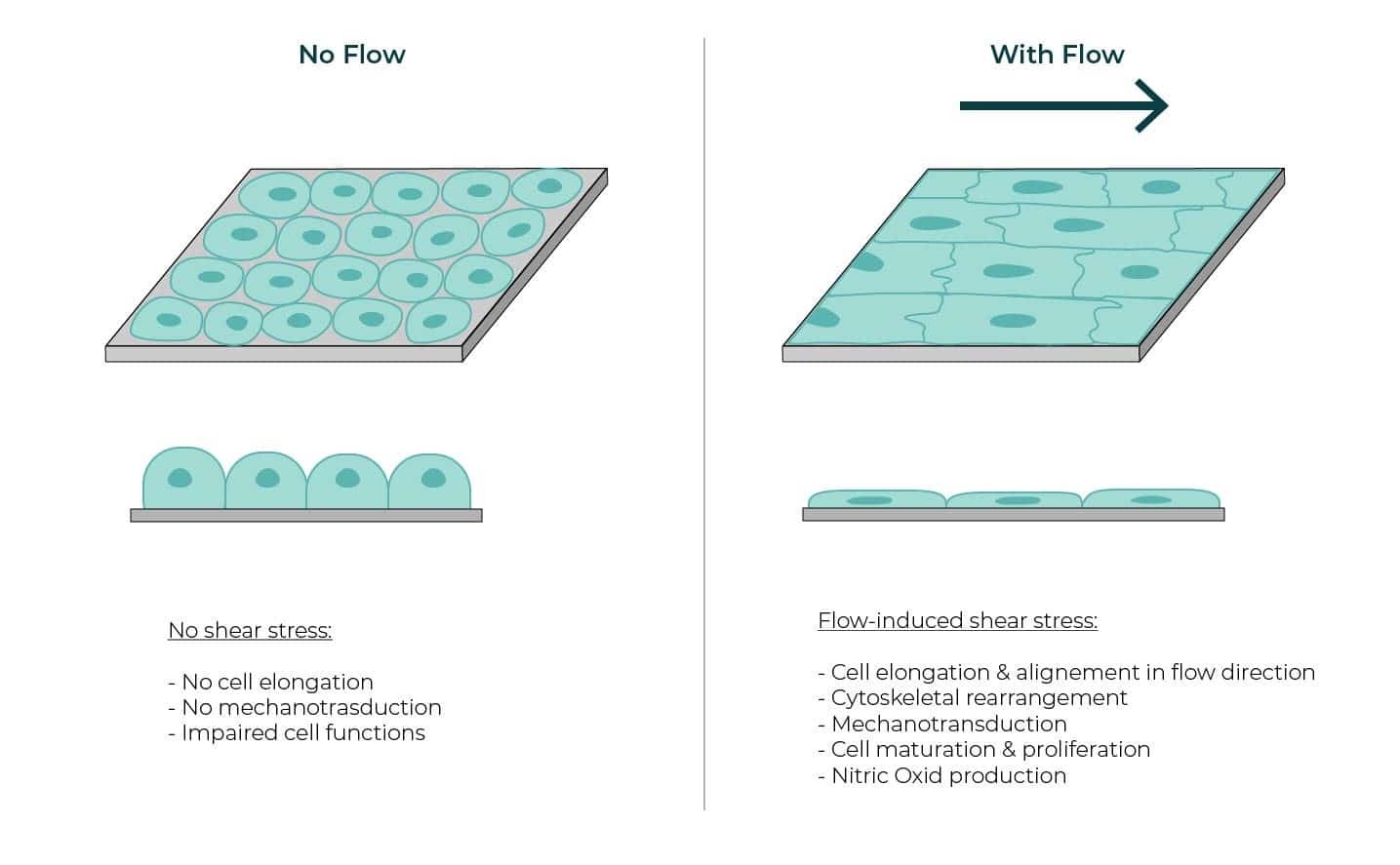
Microfluidic Cell Biology
This page is a comprehensive resource on microfluidic technologies to study cellular processes and functions. Here you can access our expertise pages, reviews to learn more about the advantages, challenges, applications, and latest developments of microfluidic cell biology.