Microfluidic recirculation system
A controlled pressure recirculation system is needed to control shear stress while continuously circulating a reagent or buffer when using a microchip or chamber. Discover Fluigent’s new microfluidic recirculation technology currently integrated into one of our products: Omi. Our compact, automated microfluidic recirculation system is a trusted solution. It is compatible with several technologies and can be adapted to be integrated into your custom project.
Benefit from the best performance and Fluigent’s expertise for your project
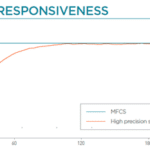
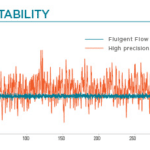
- High performance: pressure-based flow control allows for highly responsive and stable pulseless flow
- Versatility: perform any protocol including injection, uni-directional recirculation, sampling, or perfusion
- Transportability: compactly packaged, the pressure source, pressure controllers, flow rate sensors, and fluidic paths – the entire unit can be customized with injection molding techniques
- Sterility: does not require users to manipulate the chip or the system
Microfluidic recirculation system: technology description
Pressure-based flow control for stable pulseless recirculation
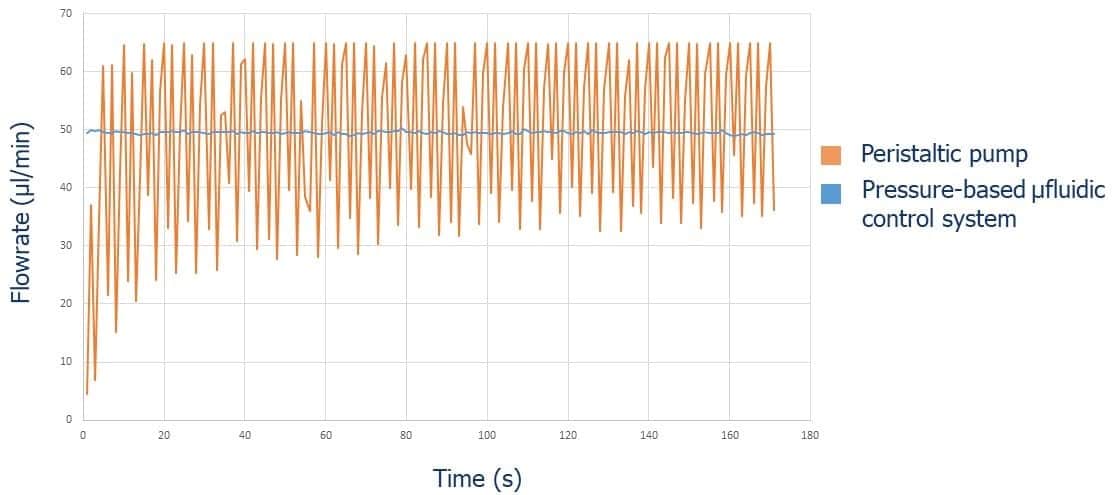
Recirculation may be accomplished with a peristaltic pump or pressure controller. The latter has the advantage of providing a more accurate and stable flow profile. The flow from the peristaltic pump leads to pulsatile flow rates with oscillations of more than 40% of the set value (50 µL/min, see graph below). Fluigent pressure controllers have a negligible flow variation with improved response time. Using a peristaltic pump can increase the risk of cell detachment, loss of membrane integrity, and/or cell dysfunction due to pulsation. Pressure-based flow controllers offer minimal shear stress comparable to that in an in vivo environment.
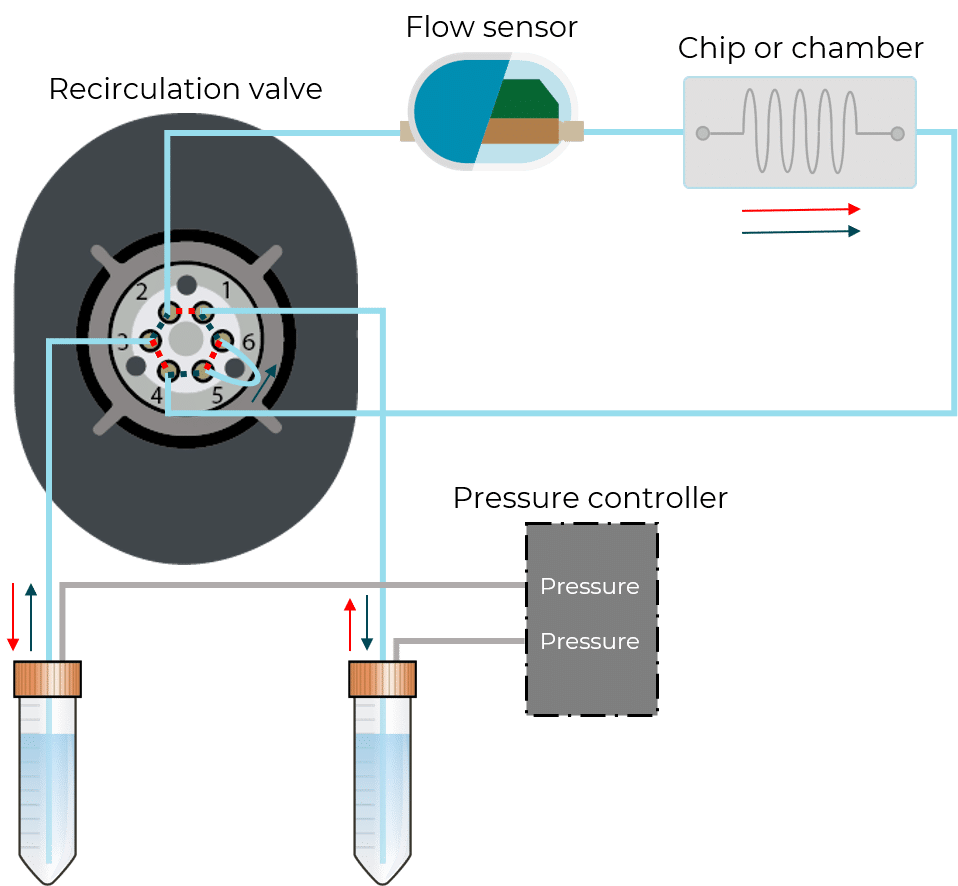
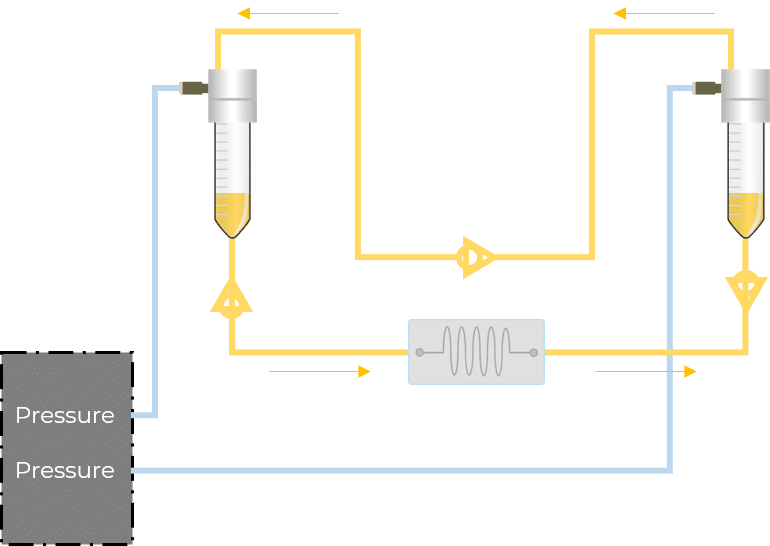
For microfluidic recirculation using pressure-based flow control, one requires a pressure source, a pressure controller with 2 channels, a 2-position 6-port valve, and a flow rate sensor. This allows for flow between two reservoirs while maintaining a continuous unidirectional flow rate in the chamber.
Although this solution is functional and offers highly improved performance, additional efforts should be made in order to mitigate the limitations of using such systems compared to a peristaltic pump, including the additional liquid volume required for this setup, the complexity and footprint, and the overall system cost.
Fluigent’s new technology allows for automatic refills so that perfusion can be performed continuously
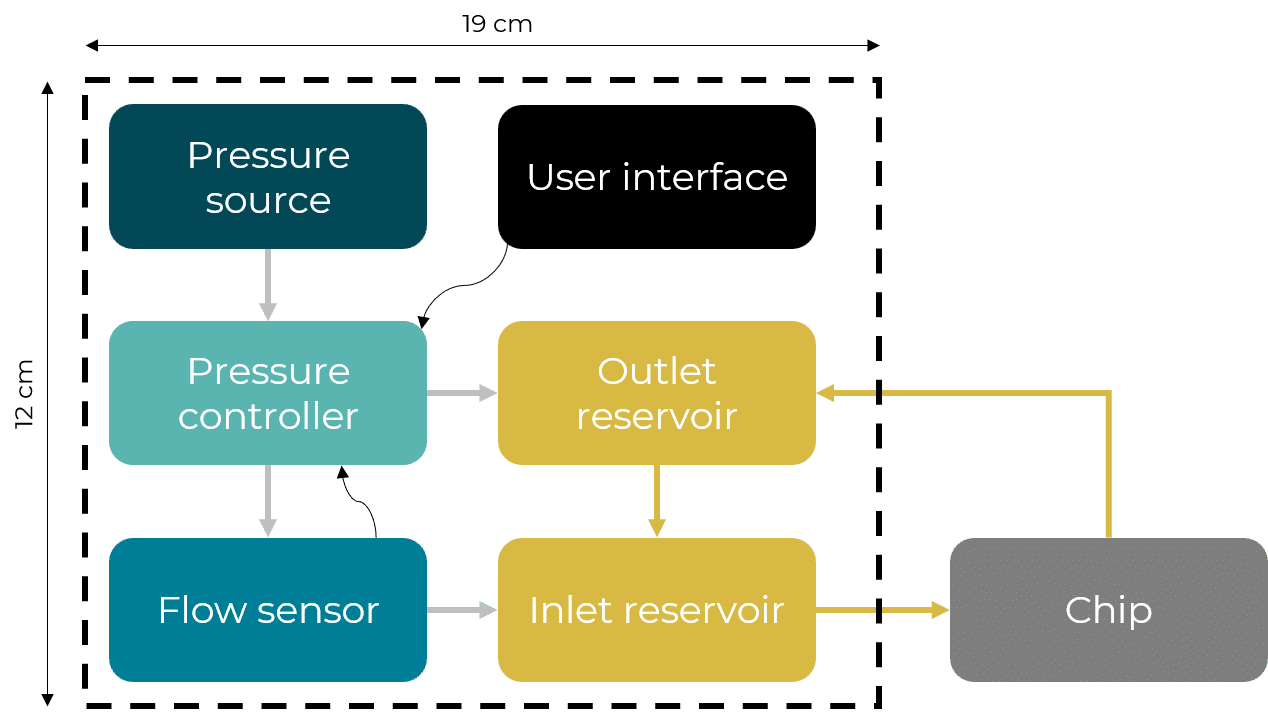
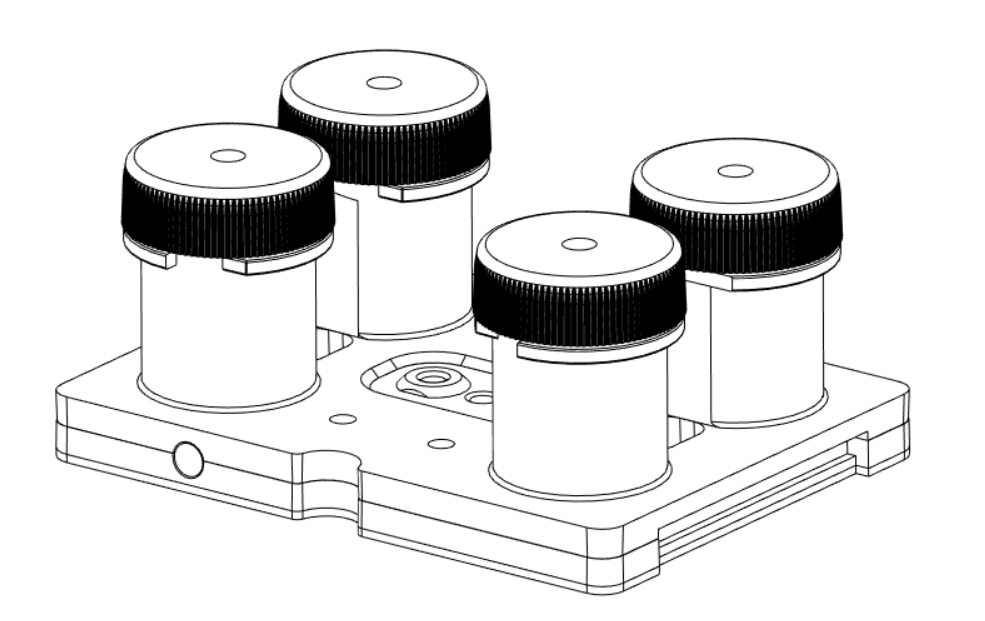
The microfluidic recirculation system directly monitors the volume left in the reservoirs and integrates all components needed in a functional microfluidic recirculation system in one compact device (that can be used with a microscope or in an incubator): pressure source, pressure-based flow controller, flow sensor (for flow rate regulation), reservoirs, all the tubing and electrical connections necessary, and a user interface (touchscreen). Users only have to provide the microfluidic module with the needed chip for their application.
Figure 2: Recirculation classical setup vs Fluigent all-integrated system
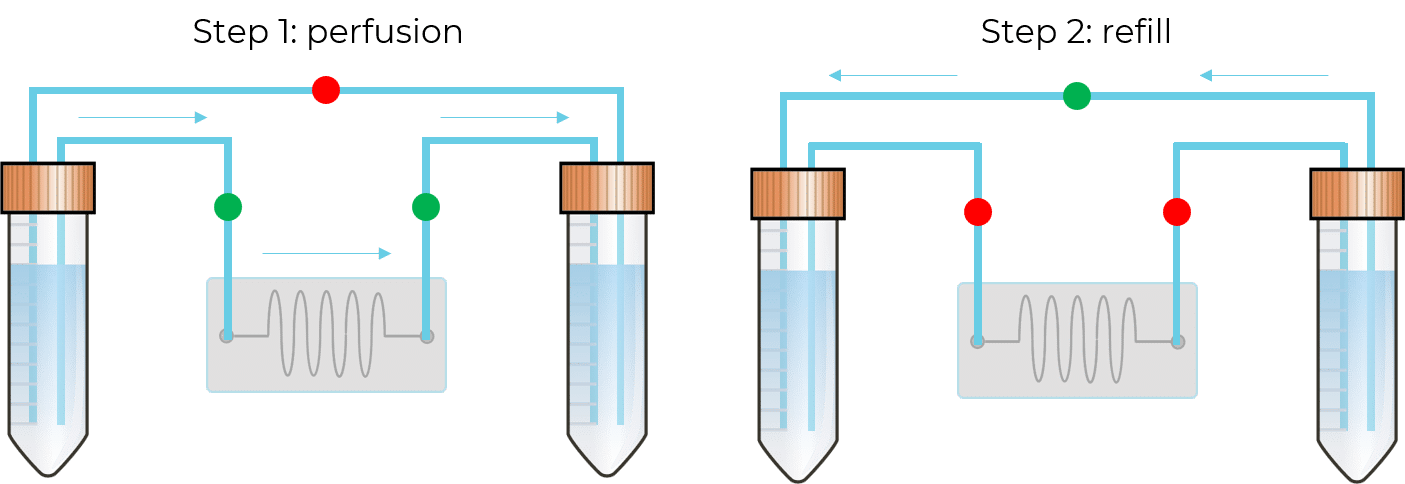
In this configuration, there is no need for a 2-position valve. Instead, there are two fluidic paths: one that goes through the chip or chamber, and one that directly links the reservoirs. In each path, the flow is feeding through in one direction with the use of valves. These enable the system to have flow going from one reservoir to the other only when required.
Optical detection for long-term flow rate reliability
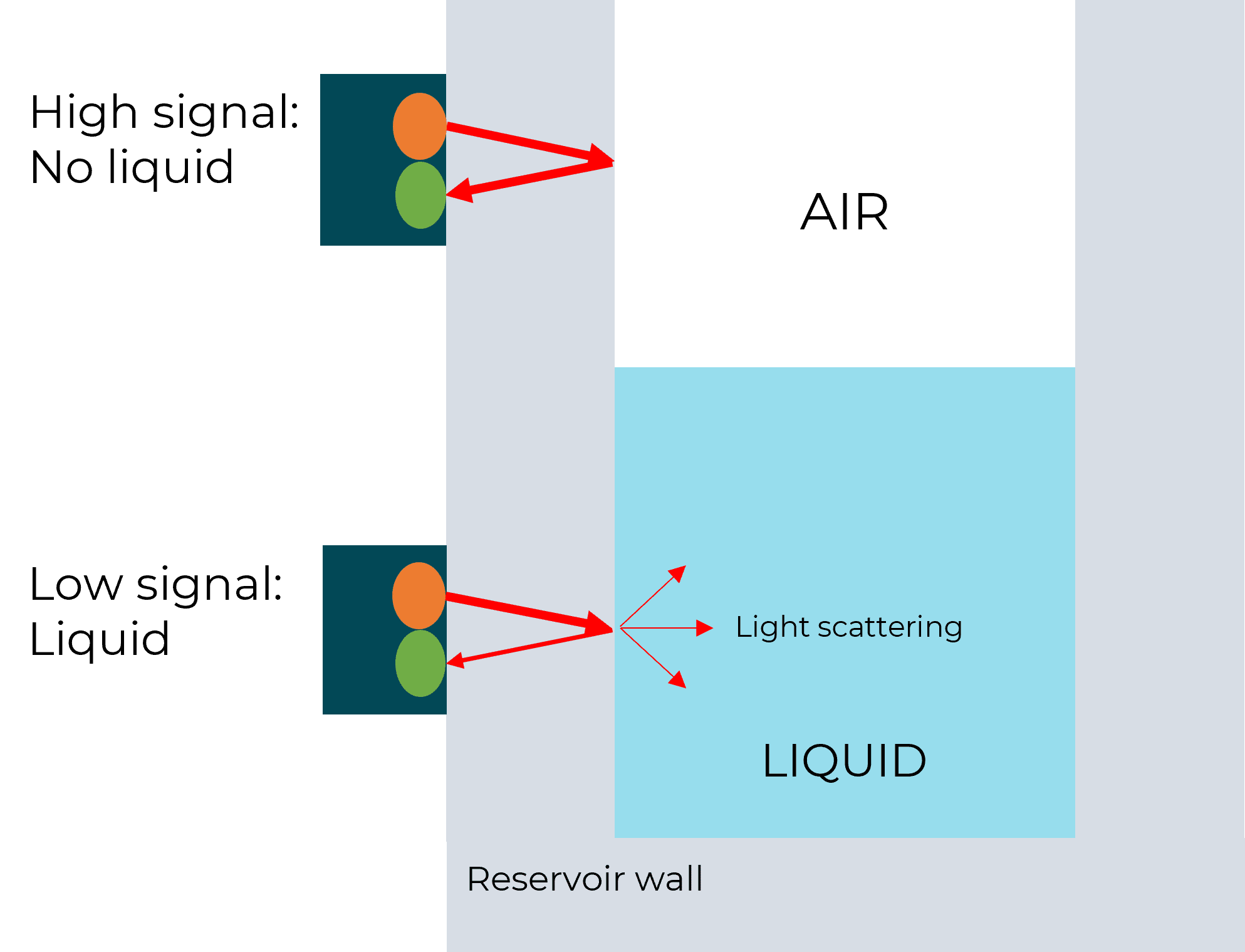
To automate the recirculation process, the device requires the ability to know the liquid level when the inlet reservoir is almost empty, or the outlet reservoir is almost full. The solution, developed by Fluigent, is based on optical level sensors (patented), it detects the presence or the absence of liquid in the reservoir.
One, or several sensors can be mounted at different heights to detect the level of liquid for which the reservoir is considered “in need of refill”. Fluigent algorithms then adjust the pressure applied to the outlet recirculation reservoir to reach the check valve’s threshold and ensure a fast refill from the outlet reservoir to the inlet reservoir while interrupting the pressure applied to the inlet reservoir for this short while.
The liquid volumes measured by the optical sensors allow users to ensure the correct flow rates are measured with the flow sensors. The volume of solution in the reservoir, according to the measured flow rate and elapsed time, is compared to the information given by the flow sensor. If a drift is detected, the flow sensor will be automatically recalibrated.
Unpluggable cartridge
Fluigent developed disposable cartridges to be used in microfluidic recirculation systems. They allow sterility and reproducibility for biological applications that require a contamination-free environment. Fluigent can also provide disposable and micro-patterned polymer-based fluidic parts that can be packaged and sterilized for medical applications.
The fabrication technology consists of a multiplayer bonding of plastic parts that can be either molded or machined to include valves to prevent backflow in the recirculation path (patented).
These components are easily integrated into a larger module with a one-click interconnection and locking technology (patented). Through an ingenious system,the cartridge can be placed and locked in its position or unlocked and removed easily. When the cartridge is in place, it is leakly-tight connected to the base manifold. Thus, neither air nor liquid can flow throughout the gasket.

Automation and software integration
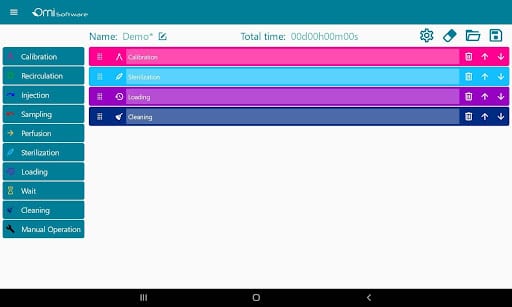
The software can manage several recirculation modules from individual users and teams.
Users can directly write protocols via the interface as it is equipped with predefined steps such as injection, perfusion, sampling, or recirculation that help users build protocols. These steps are editable, and users have the ability to choose parameters such as volumes, flow rates, durations, or flow patterns.
The data can be visualized and analyzed via graphs in real time and afterward.
Fluigent developed a solution for remote control of the microfluidic recirculation system using WI-FI connectivity. It is possible for us to design a custom Android app to edit protocols, pair, and register devices, monitor experiments, and visualize results. These functionalities can also be implemented in a web interface so that it is reachable from any device that supports a browser.
For those who prefer on-site control or want more control, Fluigent can make an embedded touchscreen included in the microfluidic recirculation system with a dedicated interface with access to basic controls.
As for data management, Fluigent provides a cloud service to collect and store data that can be retrieved when needed.
Performance of Fluigent microfluidic recirculation system
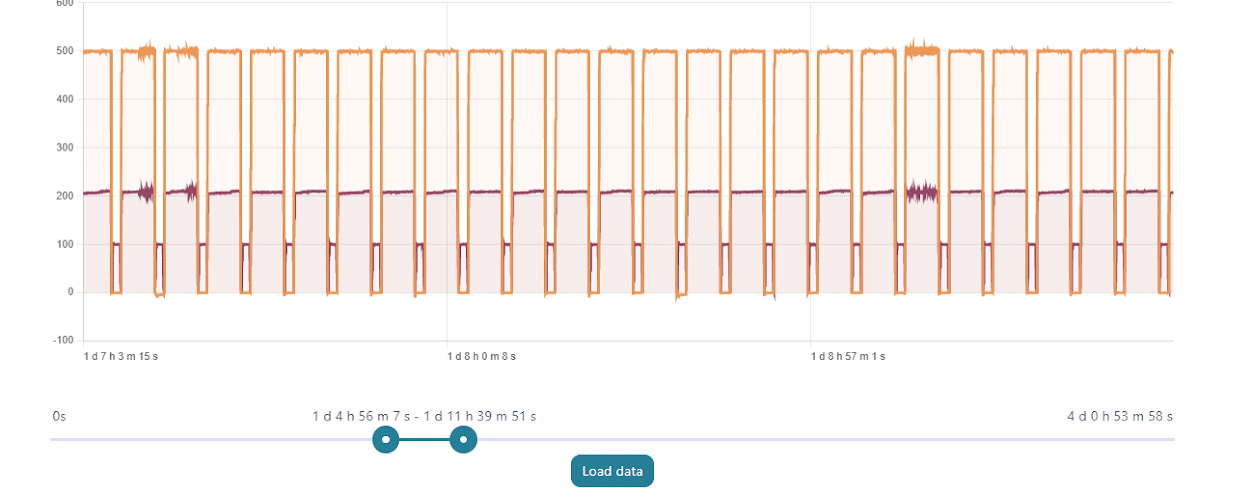
With a set perfusion flow rate of 500 µL/min, the recirculation module can maintain a constant flow rate with regular refill steps for days without any offset or issue related to the emptying of the reservoirs. On the graph above, one can see the evolution of the flow rate during 7-hour portions of a 4-day experiment.
Microfluidic applications related to microfluidic recirculation system
Dynamic cell culture
Long-term culture of cells requires the continuous perfusion of medium and thus recirculation in the fluidic path. It is interesting to have an automated and maintenance-free solution for days-long or weeks-long processes. This process must be efficient to guarantee the conservation of cells in an environment with the right amount of reagents for the entire duration of the manipulation. [1][2][3]
Live cell imaging and bioanalysis
To optically analyze live cells, scientists must keep the cells in given physiological conditions during the analysis while minimizing manual manipulations required for reagent injection, media exchange, or sample collection. Therefore, they need a module that is compact enough to be compatible with screening devices that also integrates a cell media microfluidic recirculation system to maintain the cells in the desired state over the longer term. [3][4][5]
Organ-on-a-chip
The combination of a microfluidic recirculation system with microfluidic devices can model liquid/liquid or liquid/air interfaces and mimic physiological behaviors of different human organs such as gut, lung, or heart cells inside a microchip, which is useful for drug discovery and diagnostics.
- For organ-on-a-chip-based drug discovery, recirculating media allows continuous perfusion that reproduces natural phenomena in the human body. When looking for treatment against diseases, the technology is useful to study the organs’ response to drugs in the preclinical stage to identify and validate them and prevent clinical failures later on. [6][7]
- Organ-on-a-chip development also paves the way for applications related to immunoassays, nucleic acid, or cell testing with automation, parallelization, and miniaturization of the processes. This is beneficial for point-of-care diagnostics, as the use of microfluidic recirculation permits rapid biomarker detection and thus, diagnosis of infectious diseases from sera extracted from blood with reduced costs. [8][9]
Related products
Omi, automated organ-on-chip platform
Innovative Organ on A Chip Platform
See the offerFully Custom Microfluidic Device
Fully Custom Microfluidic Device
See the offerMicrofluidic OEM Pressure Controller
OEM Fluigent PX
See the offerMicrofluidic OEM Flow Sensor
FS Series
See the offer
Ressources
References
[1] A. R. Dixon, S. Rajan, C.-H. Kuo, T. Bersano, R. Wold, N. Futai, S. Takayama, G. Mehta, “Microfluidic device capable of medium recirculation for non-adherent cell culture”, Biomicrofluidics, 2014
[2] K. B. Chang, A.-S. Kameel, Y.-K. Cho, T. Shuichi, “Pumps for microfluidic cell culture”, Electrophoresis, 2014
[3] N. Futai, W. Gu, J. W. Song, S. Takayama, “Handheld recirculation system and customized media for microfluidic cell culture”, Lab Chip, 2006
[4] C. M. Puleo, H. C. Yeh, K. J. Liu, T. H. Wang, “Coupling confocal fluorescence detection and recirculating microfluidic control for single particle analysis in discrete nanoliter volumes”, Lab Chip, 2008
[5] F. Cantoni, G. Werr, L. Barbe, A. M. Porras, M. Tenje, “A microfluidic chip carrier including temperature control and perfusion system for long-term cell imaging”, HarwareX, 2021
[6] D. R. Taft, “The Isolated Perfused Rat Kidney Model: A Useful Tool for Drug Discovery and Development”, Current Drug Discovery Technologies, 2004
[7] E. W. Esch, A. Bahinski, D. Huh, “Organs-on-chips at the frontier of drug discovery”, Nature Reviews Drug Discovery, 2015
[8] J. Ducrée, “Next-generation microfluidic lab-on-a-chip platforms for point-of-care diagnostics and systems biology”, Procedia Chemistry, 2009
[9] N. Garg, D. Vallejo, D. Boyle, I. Nanayakkara, A. Teng, J. Pablo, X. Liang, D. Camerini, A. P. Lee, P. Felgner, “Integrated On-Chip Microfluidic Immunoassay for Rapid Biomarker Detection”, Procedia Engineering, 2016