INDEX | H2020 European project
Introduction
INDEX: Integrated nanoparticle isolation and detection system for complete on-chip analysis of exosomes
The aim of the European Union’s Horizon H2020 project, INDEX (http://www.indexproject.eu/) is to isolate and characterize exosomes available in body fluids through development and integration of novel technological breakthroughs. Index gathers 6 partners from all around the world: National research Council of Italy (Italy), Boston University (United States), Institut Curie (France), Region Nordjylland (Denmark), Fluigent SA (France), Hansabiomed (Estonia).
Exosomes are biological vesicles (30-100 nm) known to mediate communication between cells. They hold great promise to revolutionize the standard of diagnostic and clinical care as they exert significant roles in various pathologies such as cancer, infectious and neurodegenerative diseases. They have emerged in recent years as biomarkers of the future for non-invasive disease diagnostics. However, both their isolation and characterization still remain a challenge due to their small size. To tackle this issue, INDEX aims at developing innovative microfluidic and optical methods to extract and analyse exosomes. The final objective of the project is to conceive a unique apparatus which automates the entire analytical process directly from patient samples.
In the last stage of the project, this equipment will be installed at Aalbord University Hospital and validated using patient plasma samples.
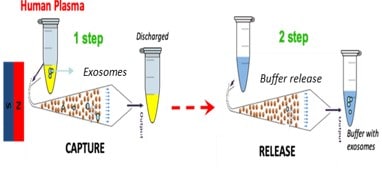
Capture and release of exosomes from human plasma.
Fluigent has the responsibility to integrate experiments performed on lab bench into a final ‘one-button operation’ instrument. This integration step is essential to make the technology accessible to non-microfluidic experts and to perform a diagnostic process in hospital. The extraction of exosomes from human plasma was the first procedure integrated into an automated microfluidic platform by Fluigent. The resulting apparatus will be presented at the end of this section.
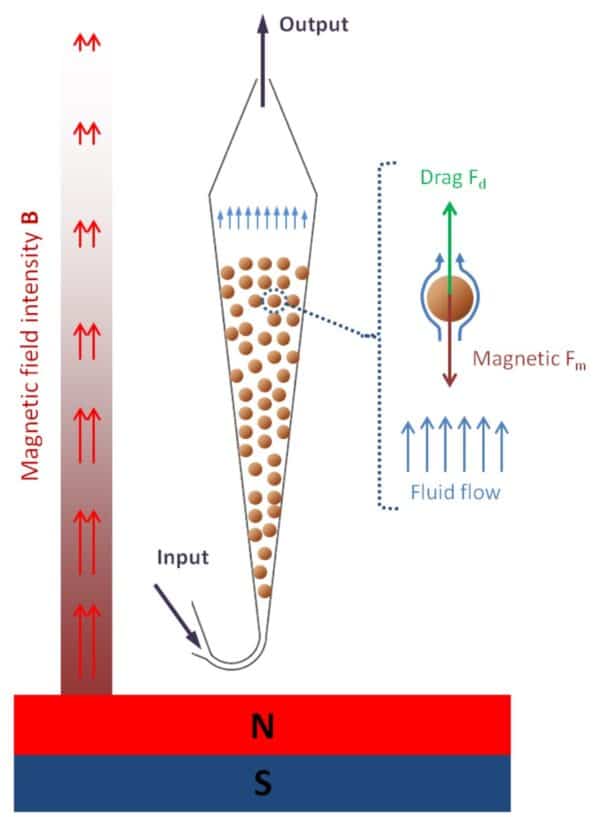
The extraction approach combines immuno-extraction and fluidized bed (FB) technology which was developed by Institut Curie (France). It involves a miniaturized FB device in which magnetic and drag forces are balanced to keep in suspension antibody-functionalized magnetic beads in a microfluidic chamber while the sample is passed through (Fig.1A). For capture, specific antibodies were grafted at the surface of magnetic beads by an original approach developed by the Institute of Chemistry of Molecular Recognition (ICRM). To capture the exosomes present in the plasma sample, the grafted beads were first introduced to the chip and the sample was then continuously injected in the FB chamber (Fig. 1B, step 1). The dynamic equilibrium created between the drag and magnetic forces allowed an effective mixing of the beads which expanded the capture efficiency by 40% compared to traditional extraction methods in tubes. Following the capture step, a specific release step was optimized to retrieve the exosomes from the functionalized beads (Fig. 1B, step 2).
The initial microfluidic platform
To perform the exosome extraction protocol described above, a microfluidic platform was installed at Institut Curie as shown in Figure 2. The set up includes up to 7 different functions (Fig. 2):
- Temperature control system
- Pressure controller
- Imaging system by using an optical microscope
- PC module with software
- Fluidized bed chip and magnet
- Flow rate controller
- Reservoirs support


The integrated system fot automate exosomes isolation

The Fluigent R&D team integrated the individual units on a single user-friendly automated prototype Fig.3). The objectives were the following:
- To make the technology accessible to untrained users
- To develop ‘a ready to use instrument’
- To reduce the total footprint of the set up
- To make the system transportable
- To facilitate the automation of protocols by the development of a dedicated and intuitive software package
Proper operation of the instrument was validated by the Institut Curie (France). The system is currently being tested at National research Council of Italy (Italy) to validate the efficiency of specific antibodies to isolate different types of exosomes. The instrument will then be installed at Aalbord University Hospital to perform diagnostic testing on patient plasma samples.
Image courtesy of Araya Farias Monica, MMBM team, Institut Curie
For more information and updates visit: www.indexproject.eu/