Droplet Digital PCR (ddPCR)
The science of microfluidic liquid handling for droplet digital PCR (ddPCR)
- Higher accuracy
- Higher sensitivity
- Absolute quantitation
What is digital PCR (dPCR)?
During the last decade digital-PCR (dPCR) has become one of the most prominent assays for analytical methods. dPCR carries out a single reaction within a sample as standard PCR, however, the sample is separated into a large number of partitions, and the reaction is carried out in each partition individually.
What is droplet digital PCR (ddPCR) and how does it work?
Droplet digital PCR relies on the partitioning of the tested sample into thousands of single samples thanks to the generation of droplets. For performing the assay, the sample volume is split in such a way that each droplet contains either one or none of the target DNA molecules. The droplets act as laboratory wells, each containing a sample, and being able to host a PCR reaction in within. Due to the small droplet volume, the PCR reaction runs very efficiently even from a single molecule. During amplification, a fluorescent dye is formed or activated. The positive droplets become fluorescent. Absolute quantitation of the number of target molecules is simplified to the count of fluorescence active droplets in the generated droplet collection. ddPCR is an excellent example of a transition of a microfluidic system from the academic field to industry.
What are the advantages of ddPCR?
The major advantage of droplet digital PCR is the possibility to perform absolute quantitation of the number of target molecules, which is simplified to the count of fluorescence active droplets in the generated droplet partition. The separation allows for more reliable collection and sensitive measurement of nucleic acid amounts. Precision is drastically enhanced [1], and no standard curves or calibration standards are necessary to assess the quantity of the sample of interest. The separation in multiple droplets containing nanoliters of reagent drastically reduces the time and reagents needed to perform a classical PCR reaction. This allows to reduce consumable and reagent costs, which makes it a one-of-a-kind cost effective method.
Main applications using droplet digital PCR technology
Liquid biopsy
Liquid biopsies are non-invasive tests performed on blood samples to detect cancer cells circulating in the blood (circulating tumor cells, CTCs) or pieces of DNA from tumor cells in the blood. This technique is increasingly used for cancer detection and monitoring, as it is low risk for the patient and helps doctors understand what kind of molecular changes are taking place in the tumor. Droplet Digital PCR (ddPC) provides the level of sensitivity required for liquid biopsy.
Copy number variation
A key measurement challenge in diagnostic research involves identifying small changes in nucleic acid sequence that are commonly associated with genetic diseases. Changes in the genomic DNA leading to an abnormal copy of a DNA sequence are called copy number variations (CNVs). They are present in complex diseases such as Down’s Syndrome and many cancers.
Pathogen Detection and Microbiome Analysis
Droplet digital PCR is extensively used in microbiology.. Digital PCR’s ability to amplify low concentration targets in complex backgrounds and show higher sensitivity than standard PCR makes it the technology choice for microbiome analysis.
Resources
Importance of fluid handling for droplet digital PCR applications
Droplet digital PCR relies on random distribution of dPCR mix containing target molecules on the partition of equivalent volumes (here, the droplets). In this way, some partitions contain no target molecules, while the remaining partitions contain at least one molecule. Partitions are next categorized and counted as positive or negative depending on their fluorescence intensity. The calculations are derived from a Poisson model, which can be impacted by partition volume since the model assumes the partition to be monodisperse. As a consequence, a heterogeneous droplet population can affect the droplet digital PCR process. In fact, several groups demonstrated through different studies that partition droplet volume variability can cause a bias on the accuracy of the measurements.
More information can be found in the paper written by Emslie et al.: Droplet Volume Variability and Impact on Digital PCR Copy Number Concentration Measurements The impact of flow rate on droplet size using a microfluidic system is today well described in the literature. Thus, to avoid heterogeneous droplet populations that can affect the droplet digital PCR process, one should consider using a precise flow controller.
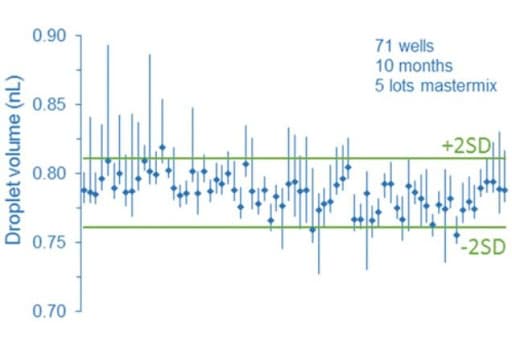

Flow control systems for industrial digital PCR
Flow rate stability is thus critical for having repeatable reactor volumes and reproducible results in droplet digital PCR experiments. Syringe pumps are commonly used for generating droplets. Depending on the model in use, syringe pumps show limited flow control. As a consequence, the droplet size, proportional to the flow rate, is affected. In addition, the actual flow rate cannot be monitored with such devices. The flow rate value is displayed on the device, but no information on the time required for reaching a set flow rate is given (the time for flow equilibrium may vary depending on the microfluidic setup, and flow can oscillate depending on the instrument). An alternative to syringe pumps is pressure-based flow controllers. These show that high-precision flow control, fast reaction time, and flow monitoring are possible.
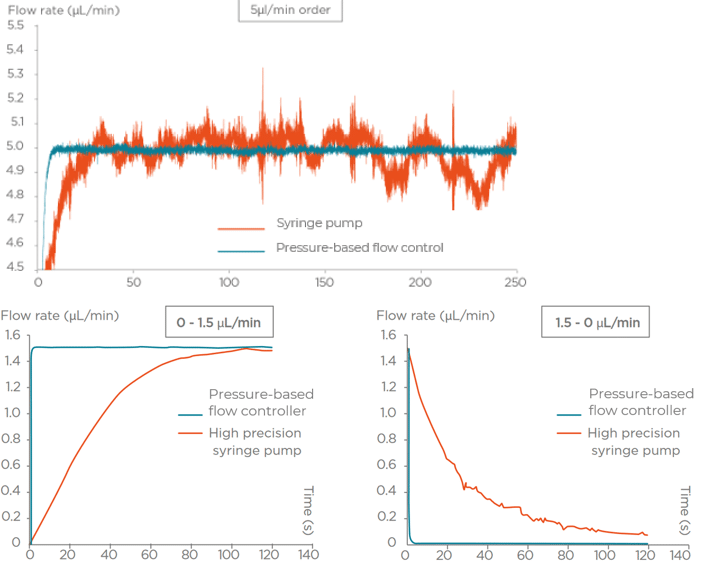

We compared the production of water-in-oil emulsions using microfluidic syringe pumps and pressure-based flow controllers. Using pressure control, the desired droplet size is quickly obtained (< 6 s), and monodisperse droplet generation is ensured over time. Thus pressure controllers are the instruments of choice for droplet digital PCR.
The benefits of choosing Fluigent for your ddPCR system
- Best in class stability: < 0.5% due to our field-proven, patented FASTAB™ technology allowing optimal flow control with the robustness required in demanding industrial environments.
- Straightforward workflow automation included in Fluigent’s software
- An expert engineering time specializing in microfluidic design and mechanical and software integration
References
1. Whale, A. S. et al. Comparison of microfluidic digital PCR and conventional quantitative PCR for measuring copy number variation. Nucleic Acids Research40, (2012).
2. Emslie, K. R. et al. Droplet volume variability and impact on digital pcr copy number concentration measurements. Analytical Chemistry91, 4124–4131 (2019).
3. Emslie, K. R. et al. Supporting information Droplet volume variability and impact on digital PCR copy number concentration measurements Author names and affiliations.
Introduction to DNA Screening Using Digital PCR
Polymerase Chain Reaction, or PCR, has an almost ubiquitous presence in biomedical sciences. Due to the ease of execution and the exquisite sensitivity offered by PCR, which covers amplification from a single template molecule up to about nine orders of magnitude, the applications of PCR are widespread. This includes targeted mutagenesis, DNA screening using digital PCR, virus diagnosis and analysis, and more. (1).
Droplet microfluidics offers significant advantages for performing high-throughput screens and sensitive assays. Droplets make it possible to considerably reduce sample volumes, which lowers costs. Compartmentalization in droplets increases assay sensitivity by increasing the effective concentration of rare species and decreasing the time required to reach detection thresholds. Droplet microfluidics combines these powerful features to enable previously inaccessible high-throughput screening applications, including single-cell and single-molecule assays (2).
Droplet digital PCR (ddPCR) is based on the amplification of single target DNA molecules in many separate droplets, which provides a new method for accurate quantification of DNA copy numbers. In a ddPCR assay, the distribution of target DNA molecules among the reactions follow Poisson statistics, which means that the majority of reactions contain either one or zero target DNA molecules. DdPCR has several advantages over more traditional qPCR methods.
- It enables the absolute quantification of target nucleic acid without the reliance on rate-based measurements and the need to use calibration curves.
- It demonstrates high sensitivity and precision for low-copy-number target nucleic acids. (2,3).
In this application note, the objective is to use Fluigent products to perform DNA screenings using digital PCR by isolating individual DNA molecules and analyzing the enzymes resulting from their expression.
How to perform high-throughput digital DNA screening
Materials: Products
Fluigent pressure-driven flow control solutions are ideal for high-throughput droplet generation for DNA screening, as the excellent flow stability offered by this technology ensures high droplet monodispersity, a controlled encapsulation rate, and experimental reproducibility.
Methods
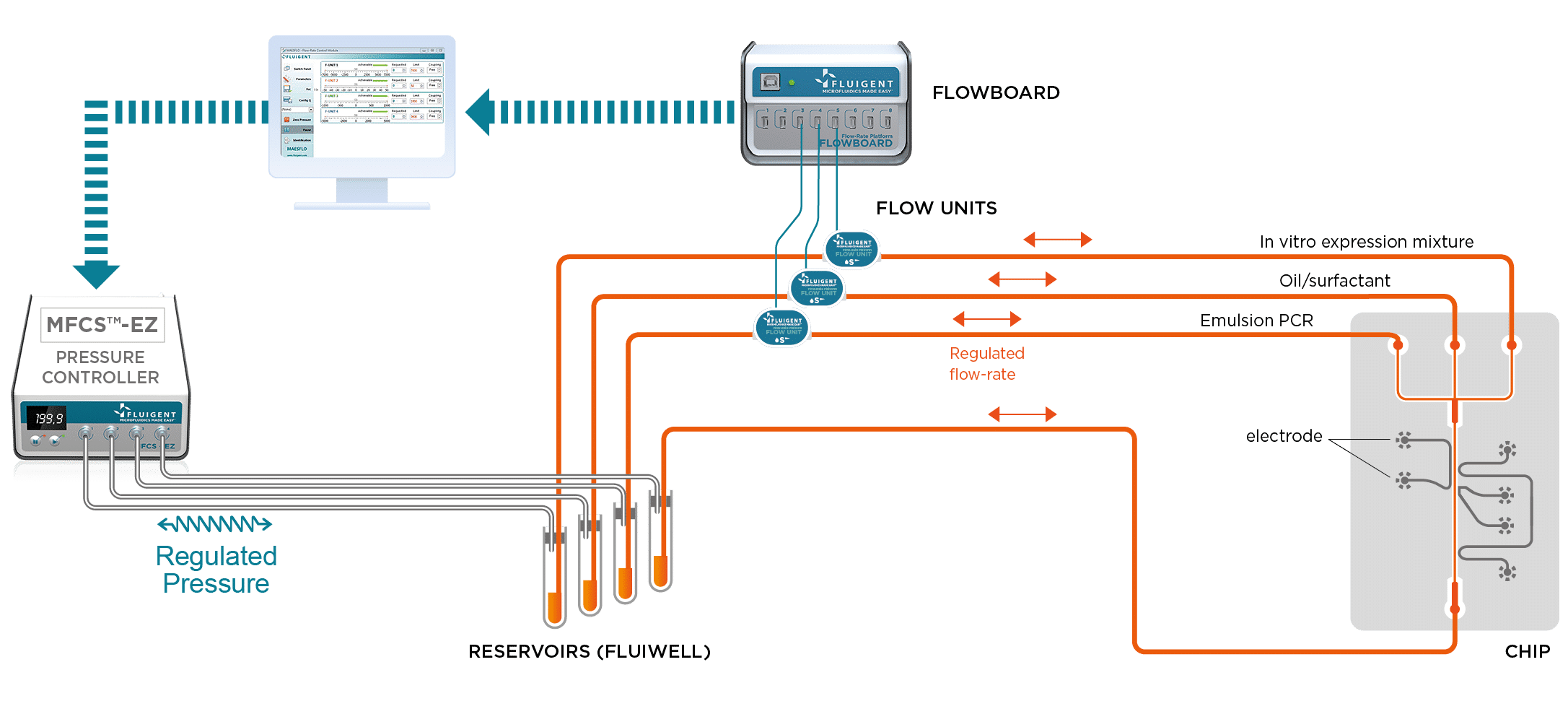
For these studies, it is important that the droplets be generated at the correct frequency and at a uniform size. Using the MFCS™-EZ in this experiment is critical, as the device generates stable flows and precise control of the different phases.
Part one: generation of the pcr/dna droplets
To perform DNA high-throughput digital screening using digital PCR, two separate emulsions were first generated. The first one encapsulated an aqueous phase of a PCR and DNA mixture in an organic continuous phase on a dedicated chip. To do this, the initial sample is injected into the microfluidic droplet generator, where it is cut into highly-monodisperse droplets by a perpendicular stream of perfluorinated oil (figure 2). Each droplet contains either one or zero molecules of DNA. The amplification step triggers the activation or production of a fluorescent dye in the droplets containing the targeted DNA. Counting the number of fluorescent droplets is equivalent to counting the number of DNA molecules.
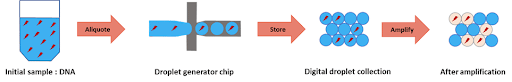
Part two: fusion of one pcr droplet with one ivt droplet generated on a chip
A green fluorescent marker is added to the PCR primers to make them visible at the end of the process. These droplets either contain DNA to be enhanced or are empty. An orange dye is also added to measure the size of the droplet, where the intensity of the light captured by a CCD camera is proportional to the size of the droplet.
For DNA screenings using digital PCR, after amplification, the PCR droplets must be injected into a second chip. This chip generates IVT droplets, while also synchronizing them to create one PCR droplet that is inserted between two IVT droplets. The IVT droplets contain an orange dye to differentiate the contents. Fusion is processed by the application of an electric field.
Results
Our set-up features fast start-up and an automated configuration that allows the experiment to be performed without constant adjustments, thus saving time and enabling systemic screening. By continuously measuring the lows and pressures, the performance of the fluidic system can be evaluated at a glance, providing full control of the process.
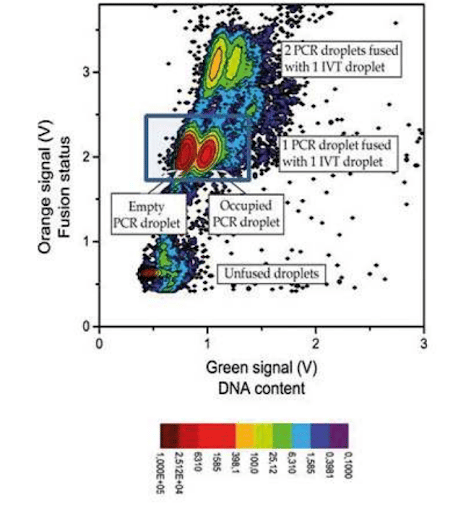
In our experimental DNA screening using digital PCR, we obtained a 1/1 fusion at an 85% to 88% success rate (no fusion: 10%, double fusion: 5%) with stable and synchronized decays.
This graph represents a count of droplets with given orange and green signals. The darker red dot signifies a large number of droplets with these characteristics, while light blue represents a low number of droplets. The green signal shows the presence of DNA in the PCR droplet, and the orange one is relevant for detecting the occurrence of fusion.
Conclusion
To perform DNA screening using digital PCR, it is essential to have high stability in order to obtain reproducible and optimal results. Flow-rate control solutions based on pressure actuation provide:
- High droplet monodispersity (even at low low-rates over long time periods.)
- Straightforward, easily automated setup
- Precise volume and low control – even with complex, multi-channel chips to minimize cross-talk between low channels.
- Ability to detect and compensate for small disruptions such as air bubbles.
Related resources
References
- Hoshino, T. and Inagaki, F. (2012) “Molecular quantification of environmental DNA using microfluidics and digital PCR,” Systematic and Applied Microbiology, 35(6), pp. 390–395. Available at: https://doi.org/10.1016/j.syapm.2012.06.006.
- Payne, E.M. et al. (2020) “High-throughput screening by droplet microfluidics: Perspective into key challenges and future prospects,” Lab on a Chip, 20(13), pp. 2247–2262. Available at: https://doi.org/10.1039/d0lc00347f.
- Markey, A.L., Mohr, S. and Day, P.J.R. (2010) “High-throughput droplet PCR,” Methods, 50(4), pp. 277–281. Available at: https://doi.org/10.1016/j.ymeth.2010.01.030.
Why use drop-based microfluidics?
Droplet-based microfluidics is a rapidly growing interdisciplinary field of research combining soft matter physics, biochemistry and microsystems engineering. Its applications range from fast analytical systems or the synthesis of advanced materials to protein crystallization and biological assays for living cells. Precise control of droplet volumes and reliable manipulation of individual droplets such as coalescence, mixing of their contents, and sorting in combination with fast analysis tools allow to perform chemical reactions inside the droplets under defined conditions.
Learn more about droplet microfluidics with this white paper
In this expertise white paper, we provide a complete overview on droplet-based microfluidics, including definitions, the reasons for using droplets (limitations of traditional methods and the emergence of new microfluidic devices), the physics behind microfluidic droplet generation, the methods of generation of different types of droplets (single emulsion, multiple emulsion…), the importance of different parameters (flow rate, surfactants…), the type of materials that can be produced, and related applications.
TABLE OF CONTENTS
I. WHAT ARE EMULSIONS AND DROPLETS?
1) Definition.
2) Applications
II. WHY USING DROPLET MICROFLUIDICS?
1) The limitations of traditional methods for droplet generation.
2) Microfluidic devices for controlled emulsions.
III. DYNAMICS OF DROPLET MICROFLUIDICS
2) Droplet formation (single emulsions)
3) Droplet generation regimes.
4) Double emulsions.
5) Complex droplet formation.
IV. IMPORTANCE OF FLOW RATE CONTROL FOR DROPLET STABILITY
V. SURFACTANTS FOR STABILIZING DROPLETS
VI. DROPLET AND PARTICLE GENERATION USING MICROFLUIDICS
1) Microparticles.
2) Nanoparticles.
3) Microcapsules.
VII. APPLICATIONS OF DROPLET-BASED MICROFLUIDICS
1) Drug delivery.
2) Diagnostics using droplet digital PCR (ddPCR)
3) Cosmetics.
References.
Download the white paper
* Required field.
All white papers
This technology can be integrated in any custom project. Users will receive the following benefits:
- Contact-free: suitable for use when sterility is required
- No calibration needed: liquid-independent
- Suitable for Industrial use: can be integrated into many devices and systems
- High precision and accuracy: Fluigent regulation control

Discover our standard OEM product based on this technology
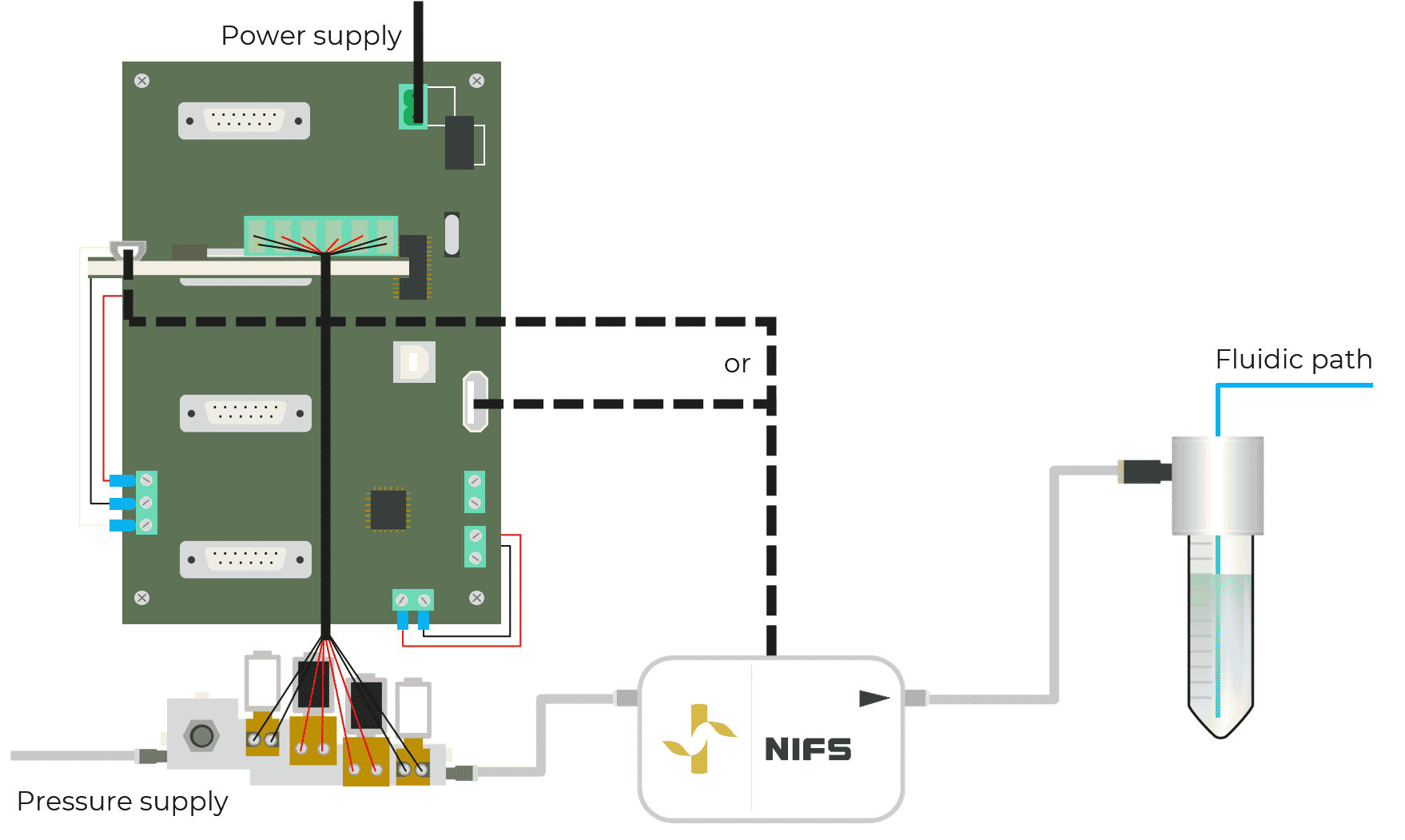
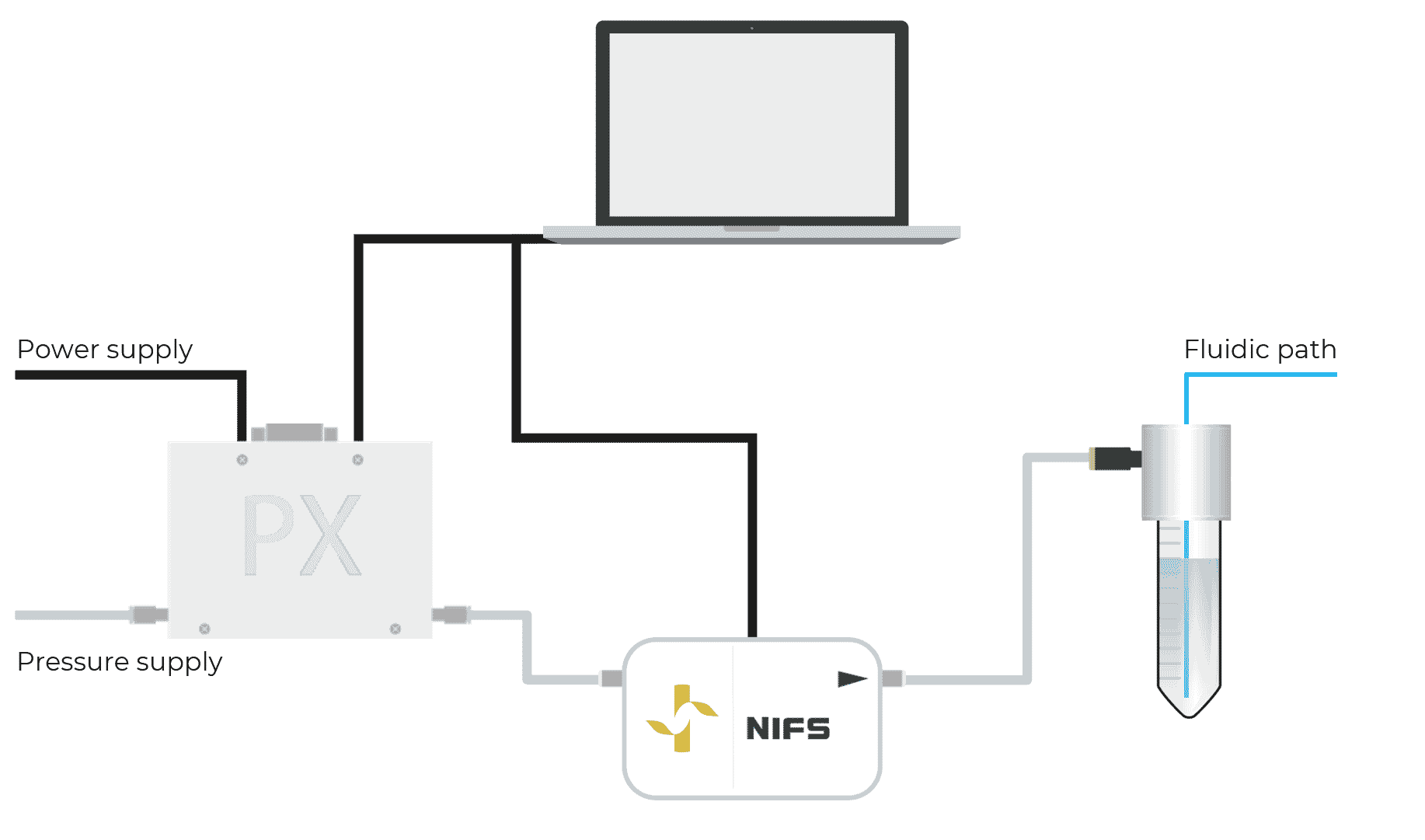
Fluigent introduced the first non-invasive flow sensor for microfluidics, the NIFS. This contamination-free Flow Sensor for industrial integration will allow you to monitor liquid flow rates and regulate them with our pressure controllers.
How does our non-intrusive liquid handling system work?
Fluigent expertise for fluid flow rate adapted to gas flow rate
Thanks to Fluigent’s expertise, including “self-learning” algorithms and calibrations, our non-intrusive flow sensing technology determines the fluid flow rate from gas flow rate measurements. Using internally developed software, the input pressure is automatically adjusted by our pressure controller to monitor or control the liquid fluid flow rate in real-time.

The technology consists of a uniquely engineered assembly of sensing, pneumatic and electronic elements that allow for precise volumetric flow rate measurement. Thanks to the conception of a zero leakage (completely sealed) system, the gas flow rate is proportional to the fluid flow rate.
No contact with the fluidic path
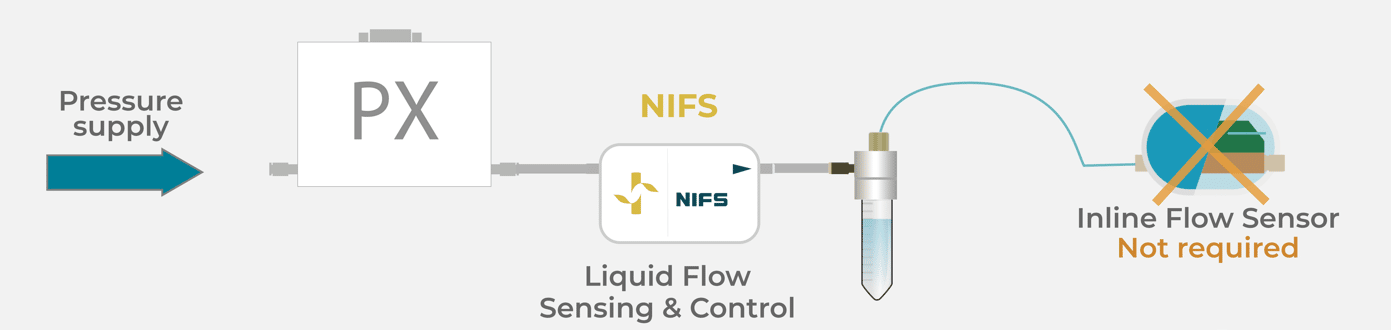
As described in figure 3, the technology is located at the pneumatic part of the system, between the regulated pressure controller and the fluidic reservoir (for more information about pressure-based flow control, see our expertise page). The flow sensing technology is never in contact with fluids.
No fluid-calibration needed
Traditional microfluidic flow sensing technologies are based on a calorimetric measurement principle and depend on fluid properties. They are generally calibrated for aqueous fluids, and additional calibrations need to be performed by the user. As explained above, with the new device, the fluid flow rate is determined by measuring the gas flow rate and is independent of the fluid properties (density, viscosity, surface tension, etc.). It makes this non-intrusive flow sensing technology universal and fluid calibration-free.
This is a technology of choice if one:
- Is working in a sterile/contamination-free environment
- Is working with several types of fluid: aqueous fluids (e.g. as water, media, PBS), blood, plasma, oils, surfactants, alcohols …
- Performs continuous operations with slow flow response time needs
A contamination-free flow sensing technology unlocking new features
PERFORMANCE
| Part Number | [INIFS] |
| Calibrated media | Air |
| Range | 100 μL/min to 10 mL/min |
| Accuracy | 5% reading |
| Lowest detectable flow increment | 5 μL/min |
| Response time | 10 s for reading 10 to 30 seconds for regulation (depends on the volume to control and the setup) |
HARDWARE SPECIFICATIONS
| Dimensions (L*W*H) | 99.85 x 45.00 x 33.50 mm |
| Weight | 117 g |
| Maximum pressure | 2 bar |
| Penumatic connector ports | 4 mm OD |
| Length of the electrical cable | 1 m |
COMPATIBILITY
| Products | FOEM: via micro-USB or via USB-A connected to computer POEM/PX: via USB-A connected to computer NOT COMPATIBLE WITH Flowboard OEM |
| Accessories | 2 mL reservoir + P-CAP screw 15 mL reservoir + P-CAP screw 50 mL reservoir + P-CAP screw |
| Product | NIFS | FS series |
| Invasiveness | Noninvasive: connected to the pneumatic path | Invasive: connected to the fluidic path |
| Calibration | Not required | Required for fluids with different viscosity than water |
| Flow rate range | 100 µL/min to 10 mL/min | 75 nL/min to 10 mL/min |
| Maximum pressure | 2 bar | 15 bar |
| Response time | 10 to 30 sec* | < 1 sec* |
| Flow stability | Excellent | Excellent |
| Recommended use | – Control of different liquid (no calibration/cleaning steps) – Long-term processes that risk contamination or clogging (non-intrusive) | – Manipulation of aqueous fluids only (no calibration step) – Processes with many flow rate changes (excellent response time and stability at low flow rates) |
*Depends on the volume to control and the setup
Applications of the non-invasive liquid handling system
Emerging applications make use of more complex fluidic operations and require compact systems. Our technology is fit for such applications as it is fully connected, compact, and provides excellent fluidic performance.
- Cell biology: biological applications such as cell culture under perfusion, immunostaining, organoid culture, organ-on-a-chip, drug discovery, single-cell analysis, cell cytometry and other applications that require a sterile environment. When using fluids such as culture media, PBS, buffers, blood, or plasma, every component in the fluidic path should be disposable, or able to be sterilized. Fluigent’s new non-intrusive flow sensing technology is ideal for such applications as it is not in contact with the fluids in play.
- Microfluidic droplet generation: When generating droplets, several types of fluids with different properties are used (aqueous solutions, oil, surfactants, alcohols). Using traditional flow sensing technology requires fluid calibration to be performed for every fluid. This step should be repeated periodically as fluid properties can differ from one batch to another. Inaccurate calibration affects the flow rate and the size of the droplets. The new sensing technology is ideal as no fluid calibration step is required, allowing one to precisely control flow rate for all types of liquids.
- (bio)chemical and molecular analysis: Chemical and molecular components can be screened using droplet microfluidic technology for drug, enzyme, or food analysis. The advantages of using this non-intrusive flow sensing technology are listed above.
- Quality monitoring and system failure prevention: flow sensors monitor fluidic protocol in real-time and warn the user of unexpected flow rate fluctuations.
Expertises & resources
Related products
Introduction to digital PCR
What is digital PCR?
Digital droplet-based assays offer promising opportunities for the absolute quantitation of low concentration analytic species. During the last decade digital-PCR assay (dPCR) became one of the most prominent assays for this class of analytical methods.
For performing the assay, the sample volume is split into multiple droplets in such a way that each droplet contains either one or none of the target DNA molecules.
Due to the small droplet volume, the PCR reaction runs very efficiently even from a single molecule.
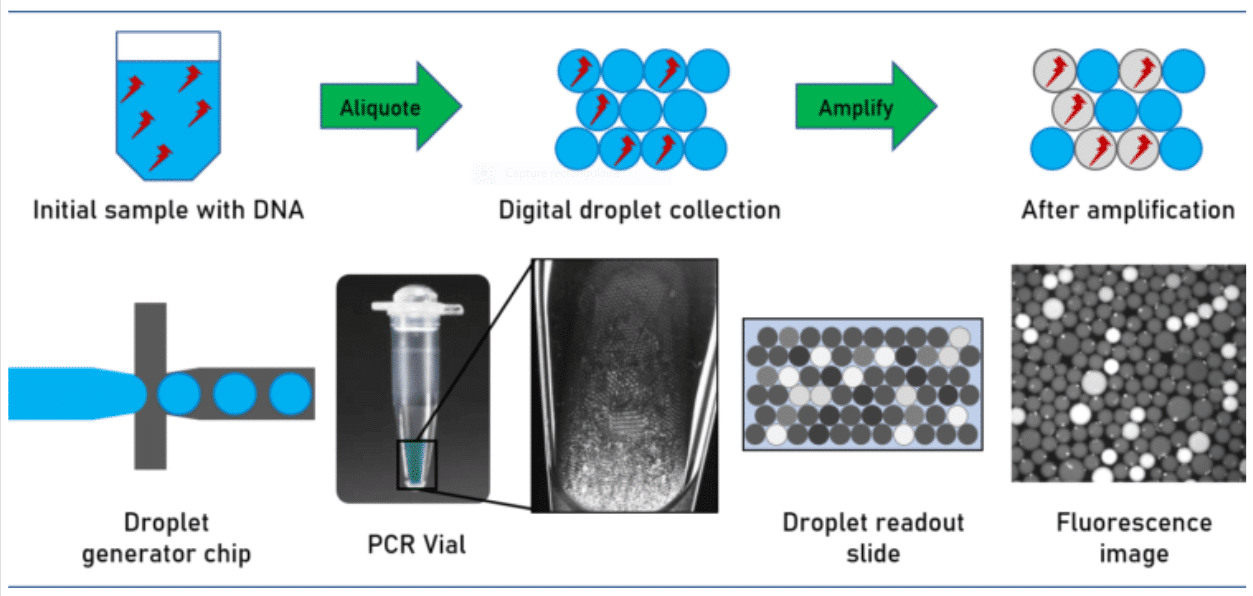
How does digital PCR work?
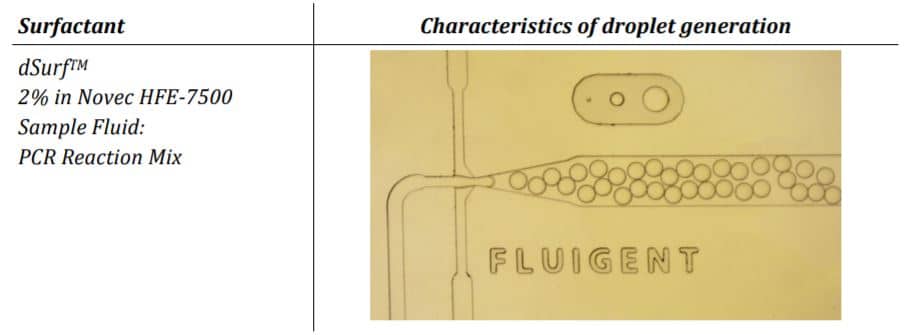
During amplification, a fluorescent dye is formed or activated. The positive droplets become fluorescent. Absolute quantitation of the number of target molecules is simplified to the count of fluorescence active droplets in the generated droplet collection. Not regarding the simplicity of the approach, its technical implementation is challenged by stabilizing the droplets collected over the complete assay avoiding unwanted droplet coalescence or crosstalk between the droplet ingredients. This has been solved by utilizing perfluorinated mineral oils as the carrier oil in combination with advanced perfluorinated surfactants, which stabilize the emulsion and avoid crosstalk and DNA exchange between the individual droplets.
In this application note we are investigating the usability of the commercially available surfactant dSurf for an exemplary digital PCR assay.
What are the materials and equipment for digital PCR experimentation?
The Fluigent droplet kit was employed for the experiments, utilizing the Fluigent EZ Drop chip with three microfluidic droplet generators per chip. Interconnection between the chip and fluid reservoirs was achieved using 2m PEEK 1/32″ tubing with an outer diameter of .010″ and two sleeves with 1/16″ outer diameter, .033″ inner diameter, and 1.6″ length. More details and dimensions of the droplet generation chip can be found on the Fluigent website.
Pressure-driven flow control was managed through the “Fluigent-MFCSTM-EZ” pressure control system. DNA amplification took place using the Eppendorf Mastercycler Gradient thermocycler. For optical readout, droplets were loaded into a disposable 10 µl cell counting chamber called “Countess™” without a grid, manufactured by EVETM NanoEnTek.
Image acquisition involved a standard fluorescence microscope (Axiovert-MAT-M, Carl Zeiss AG, Germany) equipped with a Zeiss Fluar 10x magnification NA 0.5 objective, HBO 100 light source, FITC-filter set, and an Andor-Neo sCMOS camera (Oxford Instruments, Abingdon, UK) with a 5-second exposure time for fluorescence images.

What is the method of dpcr?
Droplets were generated at a working pressure of 240 mbar for the dSurf and 140 mbar for the PCR-Mix. The chip was connected with PEEK 1/32” tubing OD x .010” and 2x sleeves 1/16” OD x.033” ID x 1.6”, tubing length: 200 mm. Generated droplets were collected into a 0.2 ml PCR vial. Amplification was performed in a conventional thermocycler with the following settings:

To acquire images, the amplified droplets were transferred to a cell counting chamber for brightfield and fluorescence imaging. The droplets needed to be arranged as a monolayer within the chamber for effective readout. This was achieved by loading 10 µl of the droplet suspension into a pipette tip and allowing the droplets to rise. The entire volume was then loaded into the chamber, starting with the pure fraction of the continuous phase to ensure the droplets were injected into the partially pre-filled chamber. After loading, the rear slit of the chamber was sealed with adhesive tape to minimize evaporation and prevent droplet motion or rearrangement during the readout process.
dPCR assay data analysis
The parameter settings of the Fluigent-MFCSTM-EZ pressure control system, as described in the Materials and Methods section, were used to generate droplets for dPCR samples. Figure 2 illustrates the observed characteristics of the generated surfactant droplets, including the droplet generation regime, size, and frequency. The average droplet size was measured to be 70 µm, with a volume of 180 pL.

Conclusion
The experiments have shown that the dSurf surfactant is suitable for scientific as well as routine digital PCR applications. The generated droplets were homogeneous in shape and size. Superior droplet stability of the dSurf surfactant system was observed during the amplification process. A few droplets have dissipated during the experiments, but this can be neglected.
The reproducibility of the experiments was also confirmed. Droplet generation with identical parameters leads to identical droplet size and quality. Summarily, dSurf can be employed as a surfactant composition for digital droplet-based assays, and therefore, for digital PCR assay.
References
1. Pohl, G. and I.-M. Shih, Principle and applications of digital PCR. Expert review of molecular diagnostics, 2004. 4(1): p. 41-47.
2. Huggett, J.F., S. Cowen, and C.A. Foy, Considerations for digital PCR as an accurate molecular diagnostic tool. Clinical chemistry, 2015. 61(1): p. 79-88.
3. Quan, P.-L., M. Sauzade, and E. Brouzes, dPCR: a technology review. Sensors, 2018. 18(4): p. 1271.
Related Resources
- 사용자 사례
Using dSurf for High Throughput Laser-Induced Fluorescence Droplet Micro-Thermometry (LuMIn)
Discover - 인터뷰 및 추천사
Testimonials dSurf
Discover - 어플리케이션 노트
E. Coli Culture in Droplets Using dSURF Fluorosurfactant
Discover - 어플리케이션 노트
Microbiome culture in droplet using dsurf surfactant
Discover - Expertise videos
MICROFLUIDICS in DROPLET DIGITAL PCR
Discover - 어플리케이션 노트
High-throughput cell DNA screening using digital PCR
Discover 
Droplet Digital PCR (ddPCR)
Discover