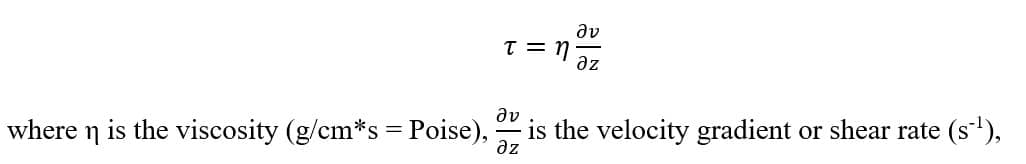Why is a controlled shear-stress a key parameter of your microfluidic experiments?
In-vivo cellular functions and morphologies are defined by several factors, which can be physical, chemical or biological. Among them, the mechanical forces constantly applied to endothelial cells have a critical impact as they are stressful and trigger cellular response. Among these forces, the shear-stress is of particular importance as it can define the cellular organization and phenotype, and the release of specific molecules that regulate the vessels’ diameter. Reproducing a controlled shear-stress in-vitro thus appears critical to better mimic the in-vivo conditions.
You can use Fluigent’s Shear Stress Calculator to determine your experimental parameters. Define the flow rate or the pressure to be applied in your system, the dimension of microfluidic chip and tubing length in order to apply the correct shear stress for your cell culture under flow conditions.
Introduction to in-vivo mechanical forces and in-vitro controlled shear-stress
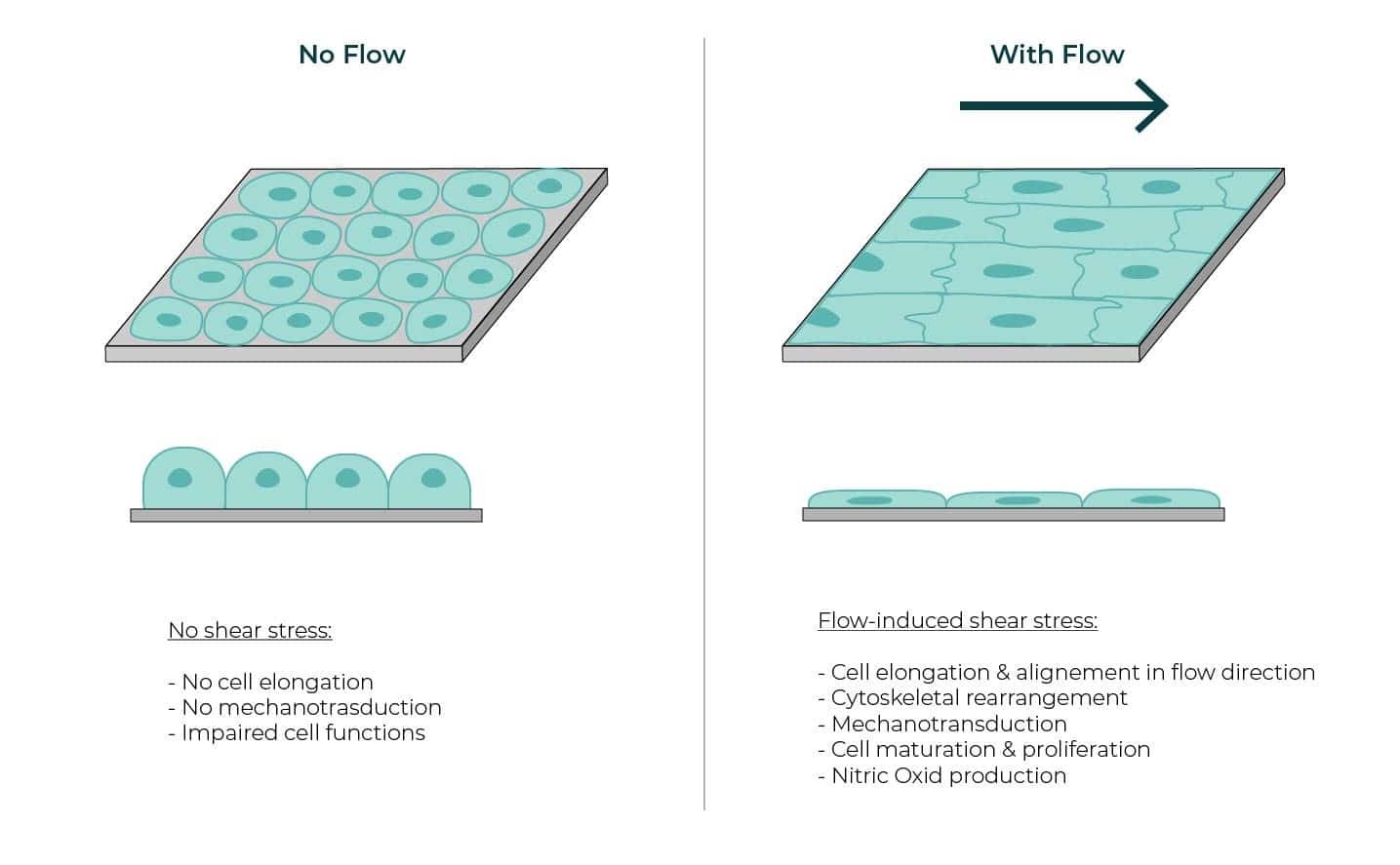
Epithelial cells (ECs), whether they are located in blood vessels, kidney or other organs such as the lymphatic system, undergo important mechanical stresses that affect their shape and function. Among these mechanical forces we can find pressure, circumferential wall, due to the heartbeat-generated pulsatile flow, and shear-stress. The latter can be defined as the tangential force of the flow acting on the epithelium. This shear-stress, as illustrated on figure 1, has a strong impact on the morphology of the cells like cytoskeletal remodeling, and can as well trigger or prevent some chemical or biological events.
In most in-vitro experiments, cells are grown on petri dishes with no application of external flow, which results in an incomplete cellular model and can lead to a false understanding of the cellular behavior. Bulk solutions were tested, for example with the orbital agitation of the culture plate, but not only were they far from the actual in-vivo condition, but they did not allow a precise control over the flow parameters.
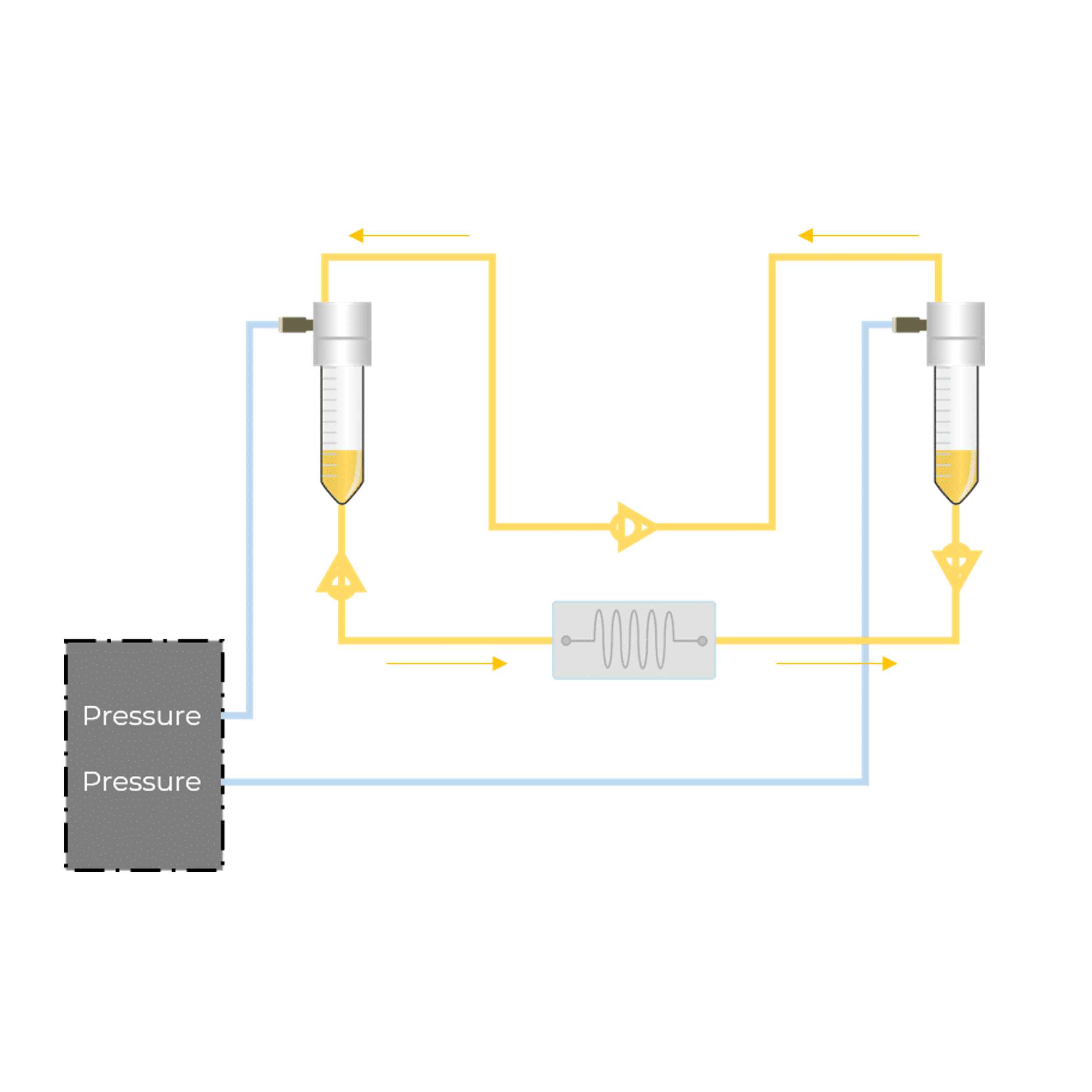
Microfluidics appears as a powerful tool to cultivate cells while perfusing them with liquid culture media, providing not only nutrients but the application of a controlled shear-stress. Indeed, the confinement inside microchannels and the laminar characteristic of the flow make it easy to reproduce the in-vessel conditions by controlling the channel dimensions and the flow-rates applied in the system.
Shear-stress definition
The physical definition of the shear-stress is the force per unit area that is created when a tangential force is applied on a surface. It is expressed as the ratio between the tangential force and the surface area, but can be simplified in the case of newtonian fluids as the following equation:
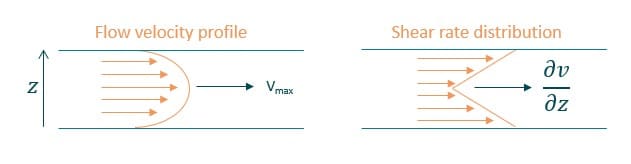
Due to the parabolic velocity profile of a laminar flow in a microfluidic channel with a maximum value in the center of the channel and a minimum value on the channel walls, the shear-stress is maximum at the walls and minimum in the center of the channel (figure 2).
Use our Shear Stress Calculator to determine your experimental parameters. Define the flow rate or the pressure to be applied in your system, the dimension of microfluidic chip and tubing length in order to apply the correct controlled shear stress for your cell culture under flow conditions.
Importance of controlling shear-stress in microfluidics application
Studying the effect of controlled shear-stress in vitro in relevant cellular systems
As previously mentioned, the lack of controlled mechanical constraints in common biological experiments prevents the generation of physiologically relevant results, which can sometimes lead to wrong or only partially true conclusions. The use of microfluidics to generate precise flows allows a fine tuning of the shear-stress applied to cells and thus study cellular behavior.
In 1981, studies were already demonstrating that the application of shear-stress of certain values could influence the endothelial cell structure and function [1]. With the development microfluidic over the past 20 years, a deeper understanding of this impact was possible.[Text Wrapping Break]Recent years saw the development of organ-on-chip models, where a functional tissue is cultured inside a microfluidic chip. It was shown that in these systems, the mechanical stimulation was crucial to regulate cell behavior and their response to infectious agents or pharmaceuticals [2].
Studying the impact of mechanical stress in bacterial biofilm formation
In recent years, the idea that mechanical shear-stress could have an impact on the development of bacterial biofilm was developed in biological studies. Comparison of the bacterial growth under various shear-stress conditions gives evidence that individual bacteria react to mechanical changes and that the biofilm shape and growth are influenced by the external mechanical forces [3]
Use our Shear Stress Calculator to determine your experimental parameters. Define the flow rate or the pressure to be applied in your system, the dimension of microfluidic chip and tubing length in order to apply the correct shear stress for your cell culture under flow conditions.
References
- Dewey C.F Jr. et al. The dynamic response of vascular endothelial cells to fluid shear stress. J Biomech Eng (1981). doi: 10.1115/1.3138276.
- Thompson et al. Mechanical Stimulation: A Crucial Element of Organ-on-Chip Models. Frontiers in Bioengineering and Biotechnology (2020). doi: 10.3389/fbioe.2020.602646
- Tsagkari et al. The role of shear dynamics in biofilm formation. npj biofilms and microbiomes (2022). doi: 10.1038/s41522-022-00300-4.
Related ressources
Microfluidic recirculation system
Read more- 微流控细胞生物学
How to reproduce active biomimetic stimulation in vitro?
Read more - 支持和工具
Shear Stress Calculator
Read more - 微流控细胞生物学
Passive and active mechanical stimulation in microfluidic systems
Read more - 实验应用说明
Development of a human gut-on-chip to assess the effect of shear stress on intestinal functions
Read more - 微流控芯片
How to choose a microfluidic chip
Read more Flow control and measurement
Read more- 微流控细胞生物学
Mimicking in-vivo environments: biochemical and biomechanical stimulation
Read more - 白皮书
Microfluidic white paper – A review of Organ on Chip Technology
Read more