什么是器官芯片?
器官芯片(OOC)技术(图1)是指使用微流控芯片内的细胞(2D或3D形式)或组织切片来复制人体器官的功能或疾病。微流控技术可以精确控制细胞环境,为细胞提供更准确的机械和生化信号(1)。这些模型支持操控少量流体,从而有利于实现可扩展的动态细胞相互作用。通过将微流控技术与OOC技术相结合,可以重现人体器官功能,以研究人体生理机能和疾病。
微流控芯片设计的最新进展是利用几何形状和结构来模拟生理条件,如长度尺度、浓度梯度和流体产生的机械力。这些仿生平台克服了传统组织培养模型面临的许多限制。
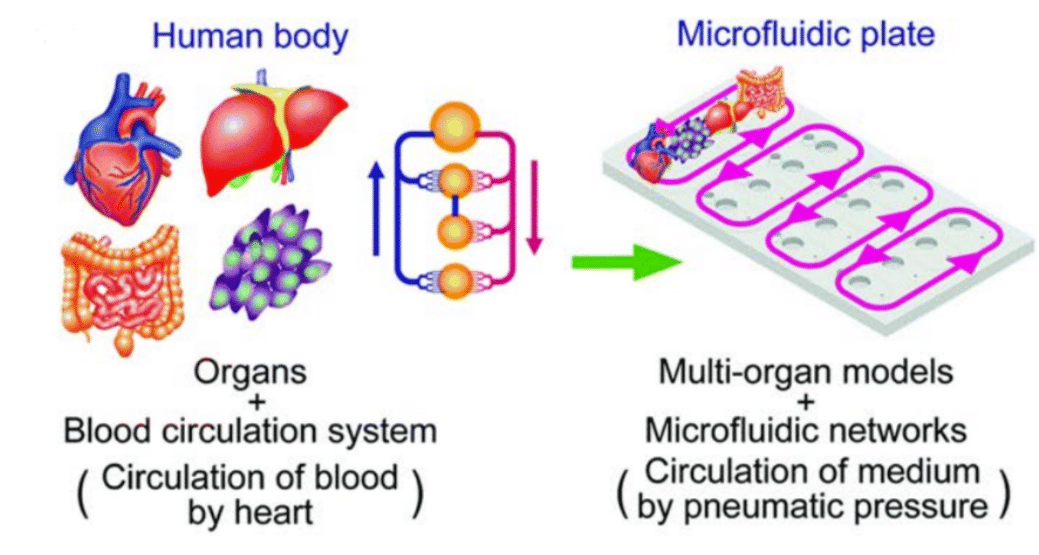
图1:从活体器官到器官芯片(1)。
器官芯片技术的应用
疗法开发
器官芯片模型通过创新的工程方法和材料为药物筛选和开发提供了卓越的多功能性。研究领域出现了一个重要趋势,即利用人诱导性多能干细胞(hiPSC)创建个性化器官模型。这些模型通过使用多孔膜将培养板连接到微流控通道,有助于探索复杂的培养设置。这种配置是研究气液界面(ALI)培养、内皮/上皮屏障和细胞间通信的理想工具。
药物发现
体外器官芯片模型的进步有望预测人类对新型候选药物的反应。这些OOC模型为精确预测和详细研究潜在药物对人类造成的毒性创造了条件。此外,它们还有助于探索新的治疗策略来对抗观察到的毒性作用。在药物发现过程中,这些模型产生的见解有助于尽早识别、修改和优化先导化合物,从而促进更安全、在临床试验中成功的可能性更高的药物开发。
个性化医疗
器官芯片模型是进行精确预测和研究潜在药物相关人类毒性的宝贵资源。它们可以深入评估不同化学物质对患者特定人体组织的影响。此外,该模型还为探索新治疗途径奠定了基础,这些治疗途径可抵消观察到的与这些化合物相关的有害影响。在药物发现过程中,从这些模型中获得的见解不仅有助于尽早识别先导化合物,而且还能对其进行修改和优化。最终,这种方法可促进更安全药物的开发,提高这些药物在严格的临床试验中成功的可能性。
适用于器官芯片应用的精确流体处理
精确的流体处理是器官芯片应用的关键要素,旨在确保准确模拟生理条件并实现各种实验设置。要在OOC应用中实现精确的流体处理,需要采用微流控组件,例如微型泵、阀和微流控通道。这些系统使研究人员能够控制流速、梯度和流体成分的动态变化,为研究器官芯片和组织芯片提供更贴近生理条件的环境。
为什么精确的流体处理在OOC模型中至关重要?
模拟生理条件
器官芯片设备需要精确控制流体流动,以准确复制人体器官的动态微环境。这种控制可确保细胞承受的流体剪切应力和梯度与人体内的细胞相似。
细胞相互作用研究和组织工程
准确的流体处理支持研究细胞相互作用,例如内皮-上皮相互作用或血脑屏障,这对于了解疾病和药物反应必不可少。其对于在芯片内创建营养物质或信号分子的梯度、促进工程组织中的组织生长和成熟也至关重要。
药物测试与开发
正确的流体处理有助于以特定浓度和速率输送药物或化合物,从而实现精确的药物测试和功效及毒性筛选。
自动化和高通量筛选
精确的流体处理系统可以实现自动化,从而支持高通量实验及化合物或条件筛选。
OOC模型示例
肺芯片 – 首个OoC
创新的人类肺泡芯片(图2)通过复制生理和病理生理综合反应重新定义了器官芯片(OoC)技术,超越了以前主要关注细胞或组织功能的模型。这一开创性的器官模型具有两个平行微通道:一个布满人肺上皮细胞,另一个布满人内皮细胞,并由微孔膜隔开。
实现细胞汇合后,将空气引入上皮层,形成气液界面,从而模拟肺泡气腔内壁。这种分隔的微型设备设计支持独立于上皮和内皮精确控制流体流动、细胞输送和营养分配。
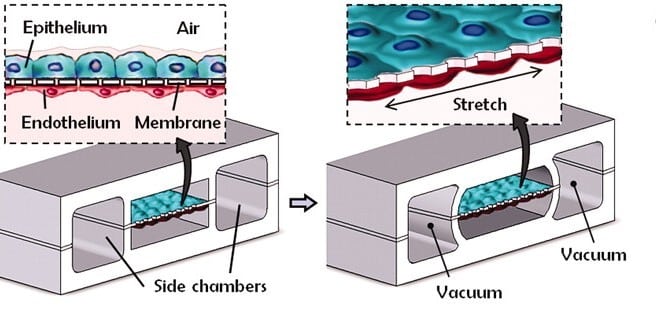
图2:受生物学启发的人肺呼吸芯片微型设备设计(2)。
肠道芯片
目前已针对小肠和大肠开发出几种创新的器官芯片(OoC)技术模型,其中利用的是带或不带底层内皮的肠上皮细胞。这些模型有两种用途:复制各种疾病并研究药物代谢和毒性。
在小肠芯片中,动态流体流动已被确定为促进绒毛形成和杯状细胞产生的关键因素,同时也促进了结肠芯片中保护性粘液层的构建。此外,模拟类似蠕动的机械运动对于实现最佳组织分化至关重要。例如,在结肠芯片模型中,循环机械拉伸和流体流动增强明显有利于细菌(以志贺氏杆菌为例)生长。
肿瘤芯片
肿瘤芯片模型正迅速成为肿瘤学研究的有力工具。这些创新系统可有效复制肿瘤微环境(TME)的关键要素,包括生化梯度、生态位因子、复杂的细胞相互作用以及由肿瘤细胞和基质细胞构成的复杂组织结构(3)。肿瘤芯片设计旨在重现组织-组织界面,并在复制癌浸润和转移过程中的复杂相互作用方面发挥关键作用。现已设计出众多模型来操控TME并探索肿瘤细胞行为,例如研究细胞对代谢梯度的反应。还有一个示例显示了使用TOC模型的意义,即用于了解肿瘤细胞在体内缺氧环境中的新陈代谢和耐药性。此创新平台有望揭示肿瘤行为的关键方面并制定克服相关挑战的策略。
Fluigent对OOC领域的贡献
Fluigent以促进科学进步为自身使命,特别是在充满活力的器官芯片技术领域。我们始终走在最前沿,不断寻求新的途径来推动研究向前发展,努力开拓创新解决方案,以重新定义科学探索的界限。
我们的承诺是推动器官芯片技术的进步,彻底改变这一领域的面貌。我们致力于寻找和提供创新解决方案,为研究人员提供支持,使他们能够更精确、更高效地探索生物系统。
References
- Wu, Q.; Liu, J.; Wang, X.; Feng, L.; Wu, J.; Zhu, X.; Wen, W.; Gong, X. Organ-on-a-chip: Recent breakthroughs and prospects. Biomed. Eng. Online 2020, 19, 9.
- Huh, D. et al. Reconstituting Organ-Level Lung Functions on a Chip. Science (1979) 328, 1662– 1668 (2010).
- Imparato, G., Urciuolo, F. & Netti, P. A. Organ on Chip Technology to Model Cancer Growth and Metastasis. Bioengineering 9, 28 (2022)